-
水是人类生存的基础性和战略性物资,而水污染则会直接影响人类的生命健康。酚类污染物是水体中常见的难降解且有毒有害的污染物之一,其中三氯苯酚(trichlorophenol,TCP)因其高毒性和致癌性已被列入中华人民共和国生态环境部、美国环境保护署和欧洲环境署发布的优先控制污染物目录。目前,处理这类污染物的方法主要有光降解法、化学还原法和吸附法等,然而,光降解法操作复杂成本高昂[1],化学还原法和吸附法处理效果较差[2],这些缺点导致它们在实际应用中受到一定程度的限制。
近年来,基于活性氧物种(reactive oxygen species,ROS)的高级氧化技术(advanced oxidation processes,AOPs)因其操作简单、反应速率高、对后续生化处理影响小等优点而受到人们的广泛关注[3-4]。传统的芬顿氧化法是目前研究与应用中最成熟的高级氧化技术之一,主要利用Fe(Ⅱ)与过氧化氢(hydrogen peroxide,H2O2)间的反应生成具有强氧化性的羟基自由基(•OH),从而对各类有机物进行有效降解[5-6]。但在均相体系中,催化剂回收利用难、反应所需pH受限、反应后产生的铁泥易造成二次污染等问题难以解决。针对这些缺陷,研究者逐渐采用含金属的非均相催化剂来替代传统金属离子进行催化反应[7-8]。其中,纳米零价铁(nanoscale zero-valent iron,nZVI)具有还原能力强,成本低,环境友好等优点,是一种理想的非均相催化剂金属[9]。然而,nZVI颗粒因其表面能高、稳定性差而易发生团聚和钝化,这导致其催化性能下降,故有必要对nZVI进行合理的修饰和改性,以提高nZVI的分散性和稳定性[10]。
近期研究表明,nZVI的硫化产物(sulfidized nanoscale zero-valent iron,S-nZVI)能有效提高nZVI的催化活性[11]。该硫化过程是在nZVI表面包裹一层硫化亚铁(FeS),FeS作为屏障保护nZVI,可以减缓nZVI的氧化速率;同时,FeS具有的低电位还可以改善nZVI的电子传输性能[12-13]。然而,在实际应用中,S-nZVI仍然存在易团聚、难回收等缺点。为此,有必要将金属负载在石墨烯、生物碳、膨润土等基体上,进一步提高其分散性和重复使用性。活性炭纤维(activated carbon fibers,ACFs)是一种高微孔碳质载体,与其它载体相比,具有较大的比表面积、丰富的反应性官能团(—COOH、C=C、C—OH和C=O等)以及廉价易回收等性质[14]。在ACFs上负载nZVI能有效提高nZVI的分散性,同时,这种块状的催化材料具有宏观立体结构,回收利用性好,在实际应用中具有独特的优势[15-18]。而协同硫化负载,将S-nZVI负载于ACFs制备得到的活性炭纤维负载硫化纳米零价铁(activated carbon fibers supported sulfidized nanoscale zero-valent iron,ACFs-S-nZVI),则不仅解决了S-nZVI易团聚、难回收的问题,还有望提升S-nZVI的循环使用性能。但迄今为止,还未有关于ACFs-S-nZVI复合材料用于去除水体中有机污染物的报道。
因此,将深入探索ACFs-S-nZVI复合材料的结构与性能之间的关系,并将其用于活化过一硫酸盐(peroxymonosulfate,PMS)降解废水中的TCP。本研究详细探究了硫铁比、反应温度、pH、催化剂和氧化剂投加量等对ACFs-S-nZVI催化性能的影响。同时,通过大量的对比实验和电子顺磁共振波谱(EPR)分析探讨了ACFs-S-nZVI活化PMS降解去除TCP的反应机理。
-
七水合硫酸亚铁(FeSO4·7H2O)、连二亚硫酸钠(Na2S2O4)和5,5-二甲基-1-吡咯啉-N-氧化物(5,5-dimethyl-1-pyrroline N-oxide,DMPO)购自上海麦克林生化科技有限公司,2,2,6,6-四甲基-4-哌啶酮(2,2,6,6-tetramethylpiperidine,TEMP)购自萨恩化学技术(上海)有限公司,硫酸(H2SO4)和硼氢化钠(NaBH4)购自永华化学股份有限公司,氢氧化钠(NaOH)和无水乙醇(C2H5OH)购自北京化工厂,商业用铁碳催化剂购自天龙净水材料厂,ACFs购自江苏苏通碳纤维有限公司,厚度为0.3~0.4 cm,比表面积为900 m2·g−1。
-
ACFs-S-nZVI等材料的制备:取一定量的ACFs浸入硝酸溶液(3 mol·L−1),然后在25 °C水浴摇床中振荡24 h,取出后用去离子水多次浸泡洗涤,置于60 ℃烘箱中干燥18 h后备用。准确称量100 mL去离子水倒入三颈烧瓶,然后在氮气环境下顺序加入1 g FeSO4·7H2O和1 g处理后的ACFs,以300 r·min−1转速搅拌2 h后,逐滴加入80 mL 1 g NaBH4和0.1 g Na2S2O4的混合溶液,继续搅拌30 min后,将取出的催化剂用去离子水和无水乙醇交替洗涤至少3次,随后置于60 ℃真空烘箱中干燥6 h。最后得到的块状黑色纤维就是S/Fe比为0.3的ACFs-S-nZVI。此外,通过相同条件制备未加Na2S2O4的ACFs-nZVI和未加ACFs的S-nZVI。
-
取20 mL浓度为10 μmol·L−1的TCP溶液放入锥形瓶中,用H2SO4和NaOH调节溶液pH,并加入一定量的PMS和ACFs-S-nZVI,然后将锥形瓶置于恒温振荡器中,调节反应温度至一定值,设置转速150 r·min−1,分别于5、10、15、20、25、30 min时取样2 mL,并经0.22 μm滤膜过滤,然后用高效液相色谱仪(HPLC)测定滤液中TCP的含量。每组实验设2个平行样。常规实验中,反应条件设置为温度25 ℃、pH=6、TCP浓度10 μmol·L−1、催化剂投加量0.5 g·L−1、PMS投加量0.5 mmol·L−1。
-
TCP浓度用液相色谱仪(Waters 2 707)测定,流动相为甲醇和去离子水溶液(V∶V=80∶20),流速0.2 mL·min−1,柱温35 ℃,进样10 µL,检测波长295 nm。溶液中的铁离子浓度用硫氰化钾(KSCN)紫外分光光度法测定,PMS浓度用碘化钾(KI)和KSCN紫外分光光度法测定。
-
采用场发射扫描电子显微镜(FE-SEM,Vltra55)和能谱仪(EDS)观察S-nZVI及ACFs-S-nZVI的形貌和元素分布。采用X粉末衍射仪(XRD,Ultima Ⅳ)对样品的晶体结构、组成进行测试。测试条件为Cu-Kα(40 kV,40 mA),额定输出功率为3 KW,扫描角度2θ为10°~80°,扫描速度为5 °·min−1。采用傅里叶变换红外光谱仪(FT-IR,Nicolet 5700)对样品进行扫描,扫描波数为600~4 000 cm−1,分辨率为4 cm−1。采用X射线光电子能谱仪(XPS,Thermo K-Alpha)对样品在反应前后表面元素组成及价态的变化进行测试及分析。
-
利用电化学工作站(CHI 604E)进行电化学测试,测试采用三电极系统,分别以铂片、饱和甘汞电极和玻碳电极作为对电极、参比电极和工作电极,所用电解液为1 mol·L−1 KOH溶液。为制得所用的ACFs-S-nZVI和ACFs-nZVI改性后的玻碳电极,取1 mg充分研磨后的催化剂均匀分散在稀的Nafion溶液(0.5%,50 µL)中,然后取24 µL(分12次,每次2 µL)混合后的溶液滴在干净电极上,空气晾干后备用。
线性扫描伏安法(LSV):搭建电化学测试装置,然后选择LSV模式,设置电位区间(3~-3 V)和扫描速率(50 mV·s−1)等参数,测试开始后电压将从起始电位向终点电位进行单向扫描,最终得到的电极电流随电极电位变化的曲线即为线性扫描伏安图。
电化学阻抗技术(EIS):搭建电化学测试装置,先确定开路电压,然后选择EIS模式将其设为初始电压,同时设置高频处频率(100 KHz)、低频处频率(1 Hz)以及振幅(0.005 V)等参数,最终得到的不同频率下阻抗的实部Z'和虚部Z''绘制的曲线即为电化学阻抗谱。
-
在1 mL反应溶液中加入适量DMPO或TEMP作为捕获剂,混合均匀,用毛细管取适量混合溶液放入电子顺磁共振波谱仪中,仪器参数设定为:中心场3 510 G,扫描宽度为80 G,微波频率为9.77 GHz,功率为20.00 mW。利用EPR鉴定ACFs-S-nZVI/PMS体系中产生的自旋加合物特征峰,从而进一步判断体系中产生的ROS。
-
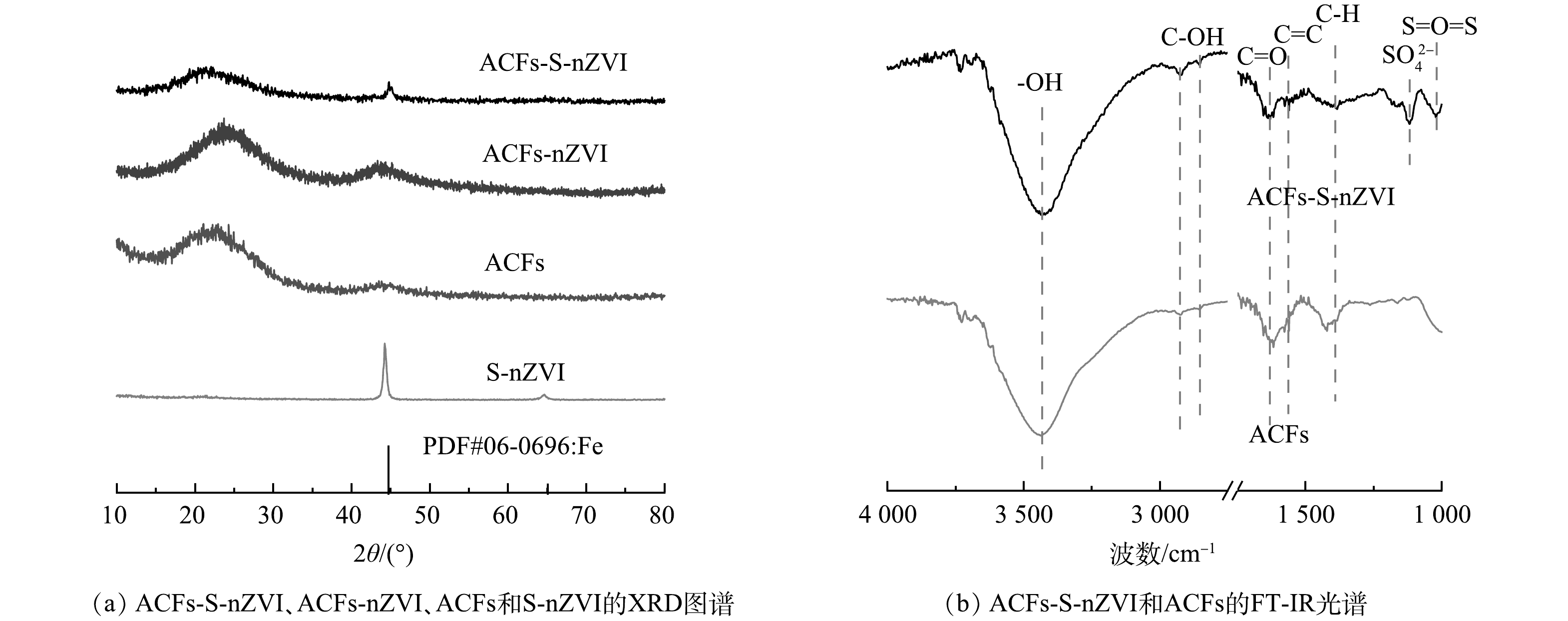
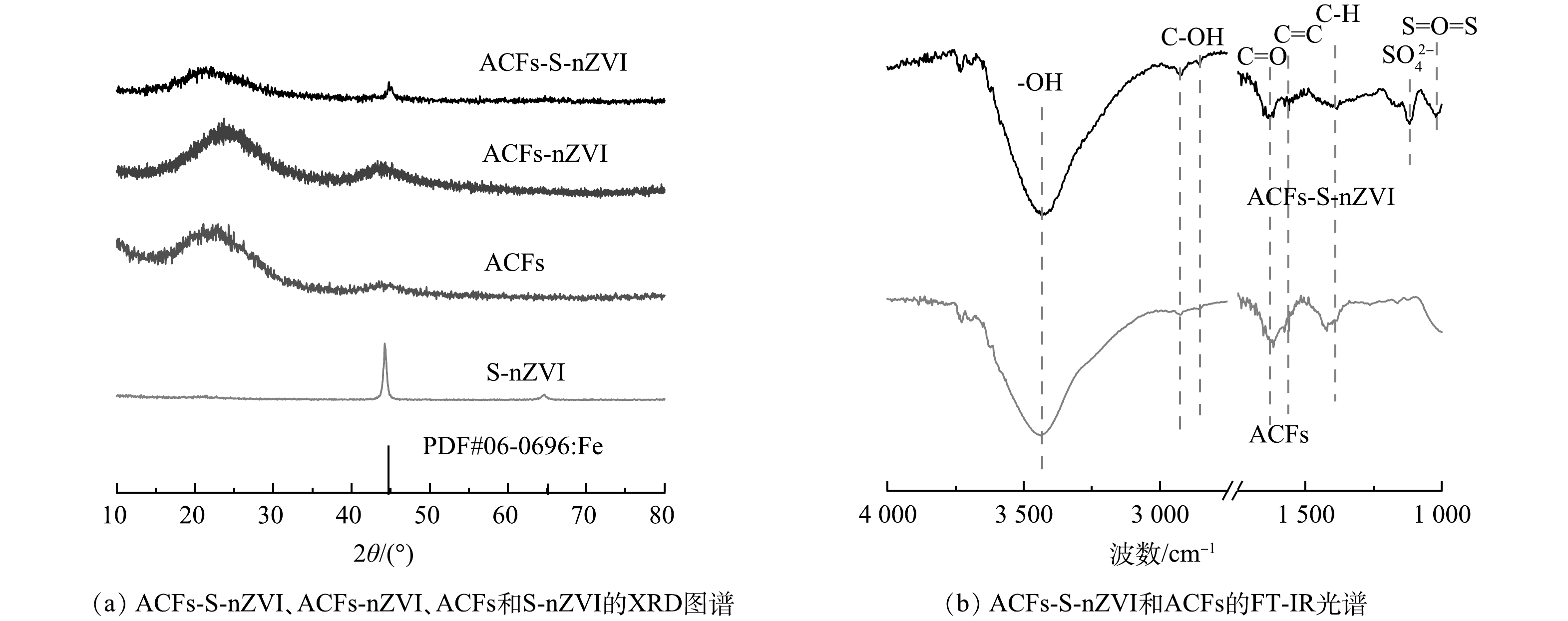
采用扫描电子显微镜(SEM)对S-nZVI、ACFs-S-nZVI的表面形貌进行分析,结果如图1所示。可见,S-nZVI颗粒呈球形链状排列(图1(a)),有明显团聚现象,颗粒直径大多在200 nm左右(图1(b))。图1(c)是在ACFs表面负载S-nZVI后的ACFs-S-nZVI图像。可见ACFs具有纤维状结构,是理想的载体材料,且ACFs表面覆盖了一层S-nZVI颗粒,颗粒相对分散和均匀(图1(d)),无明显团聚现象。值得注意的是,经负载改性后的颗粒直径明显减小,主要集中在20 nm左右,说明ACFs对S-nZVI起到了较好的分散和稳定的作用,这可归因于ACFs具有较高的比表面积、丰富的微孔结构和较多的反应性官能团,能有效吸附金属并为其提供稳定位点[19]。此外,由元素映射结果(图1(e)~(i))可见,Fe、S、C和O元素在ACFs表面均匀分布,证实了S-nZVI在纤维表面的成功合成。
采用X粉末衍射仪(XRD)对样品的晶体结构、组成进行测试,结果如图2(a)所示。可见,ACFs在2θ=22°~28°处存在无定型碳的衍射峰,负载nZVI后,ACFs-nZVI在2θ=44.6°和65.0°处出现了新的衍射峰,分别对应于α-Fe的[110]和[200]晶面,进一步证明了负载在ACFs表面的纳米粒子为Fe。值得注意的是,硫化过程中nZVI表面形成了FeS保护层,可有效抑制α-Fe的氧化,但图谱中未观察到与含硫化合物相关的峰,可能是因为其含量或结晶度较低[20]。此外,与S-nZVI材料相比,ACFs-S-nZVI材料α-Fe特征峰的衍射强度相对较弱。这是由于ACFs对S-nZVI具有分散和稳定的作用,进而限制了ACFs-S-nZVI材料表面颗粒直径的增长,降低材料的结晶度[21],这与图1(b)、图1(d)中经负载改性后的颗粒直径显著减小的现象一致。
通过傅立叶红外光谱(FT-IR)分析了ACFs-S-nZVI和ACFs的表面官能团。如图2(b)所示,在600~4 000 cm−1内,ACFs-S-nZVI和ACFs的FT-IR光谱近乎一致,均存在—OH (3 441 cm−1)、C—OH (2 858,2 932 cm−1)、C=O (1 640 cm−1)、C=C (1 557 cm−1)和C—H (1 425 cm−1)等伸缩振动。但与ACFs相比,ACFs-S-nZVI在1 118 cm−1和1 031 cm−1处出现了新的特征峰,分别对应于SO42−和S=O=S的伸缩振动,这进一步证实S-nZVI已成功负载于ACFs表面[22]。
-
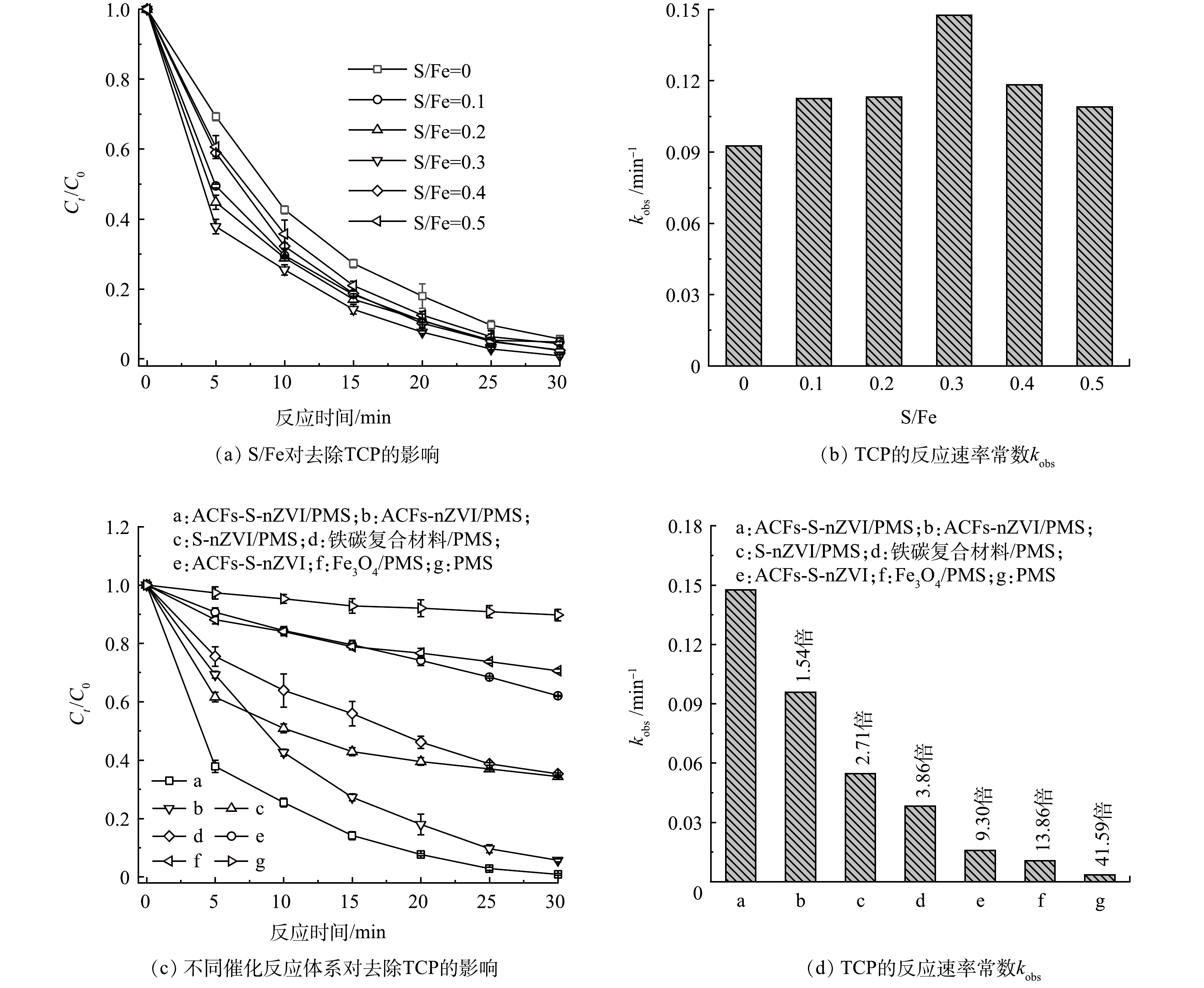
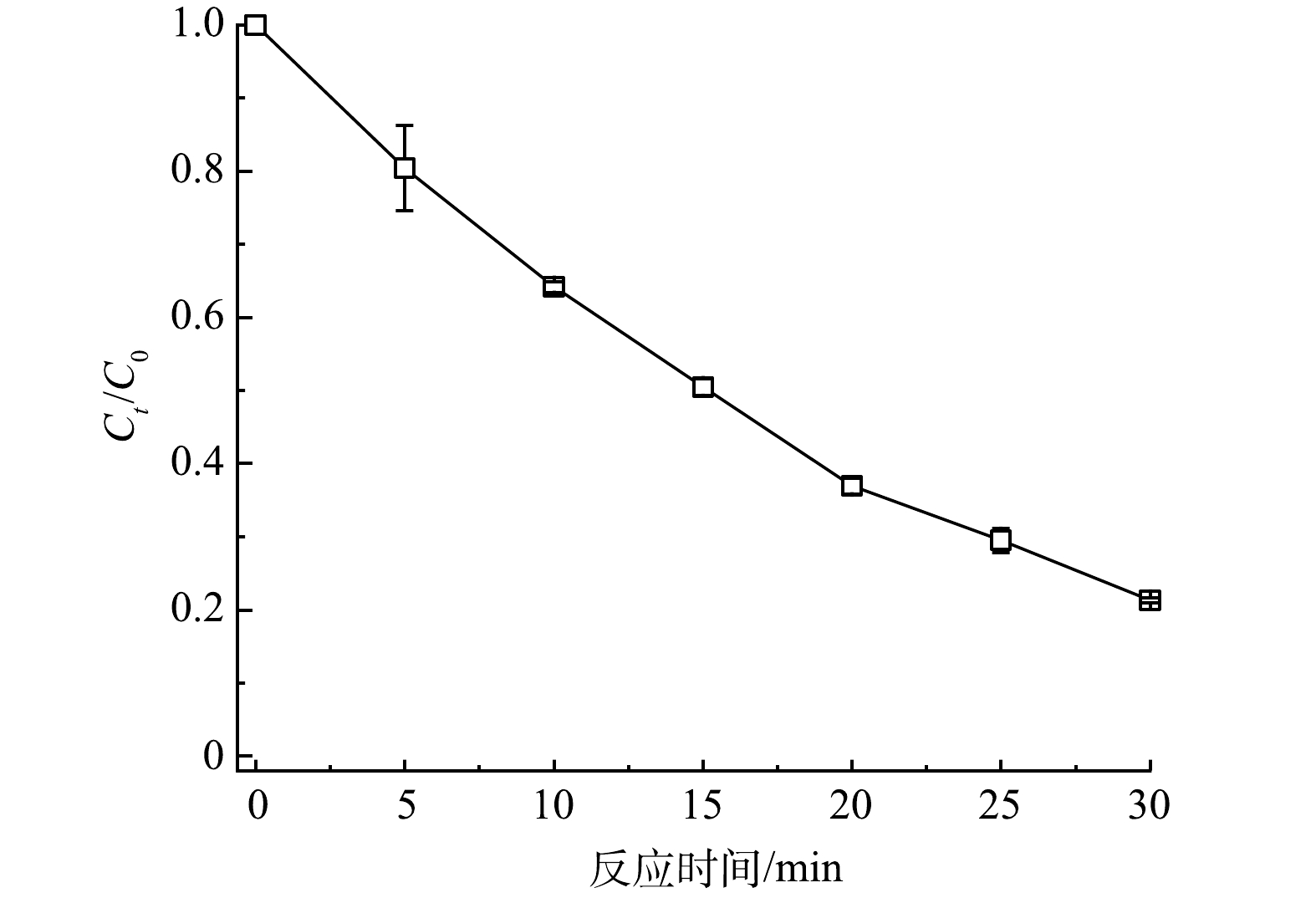
S-nZVI中硫的含量对其反应活性有着重要影响[23],因此,本研究考察了不同S/Fe比对ACFs-S-nZVI/PMS体系去除浓度为10 μmol·L−1 TCP的影响,结果如图3(a)~(b)所示。随着S/Fe比的增加,TCP的去除率和反应速率常数均呈现先升高后降低的趋势。这是因为随着硫掺杂水平的提高,nZVI表面形成了FeS,其具有比nZVI(4.06 eV)更高的负电位(5.02 eV),进而提高了体系的电子转移速率,但过度硫化会消耗大量nZVI并生成二硫化亚铁(FeS2),而FeS2带隙较高(0.95 eV),会减缓电子的传递速率,进而阻碍了TCP的去除[24]。通过对比发现,当S/Fe比为0.3时,所合成的催化剂在TCP的去除反应中呈现出了最佳的去除率(99.14%)和反应速率常数(kobs=0.148 min−1,计算方法见式(1)),因此,后续的研究主要针对S/Fe比为0.3时的ACFs-S-nZVI材料作深入的分析。
式中:t为催化反应过程中某一时刻,min;C0为污染物的初始浓度,μmol·L−1;Ct为t时刻的浓度,μmol·L−1;kobs为一级动力学速率常数,min−1。
进一步地,本研究还探究了ACFs-S-nZVI/PMS、ACFs-nZVI/PMS、S-nZVI/PMS、Iron-carbon/PMS、ACFs-S-nZVI、Fe3O4/PMS和PMS不同体系去除浓度为10 μmol·L−1 TCP的性能,如图3(c)~(d)所示。30 min内,单独的PMS和ACFs-S-nZVI体系仅能去除10.26%和37.97%的TCP,而ACFs-S-nZVI/PMS体系对TCP的去除率可达到99.14%。较为突出的是,ACFs-S-nZVI/PMS体系在5 min内即可去除62.15%的TCP,而相同时间内,S-nZVI/PMS和ACFs-nZVI/PMS体系对TCP的去除率仅为38.41%和30.72%,说明硫掺杂和以ACFs作为载体可以显著提升nZVI的催化性能,这可归因于硫化增加了nZVI的电子传递速率,而ACFs作为载体可以对nZVI起到分散和稳定的作用[25]。值得注意的是,ACFs-S-nZVI/PMS体系去除TCP的速率常数(0.148 min−1)分别约为S-nZVI/PMS体系(0.055 min−1)和ACFs-nZVI/PMS体系(0.096 min−1)的2.71和1.54倍,且其与已商业化的Iron-carbon/PMS体系(0.038 min−1)和Fe3O4/PMS体系(0.011 min−1)相比,也展现出了极大的优势,这进一步说明了ACFs-S-nZVI/PMS体系具有优越的催化性能。
-
反应温度、pH、催化剂和氧化剂用量是评价催化剂性能的必要条件[26]。如图4(a)所示,随着温度的升高,TCP(10 μmol·L−1)的去除率也逐渐增加,说明温度的升高有利于反应的进行,这是因为升高温度可以增加分子间碰撞的频率,进而加速了PMS的活化,TCP的去除率也随之提高[27]。同时,根据阿伦尼乌斯方程计算出反应的活化能(Ea)为12.03 KJ·mol−1(图4(a)插图),说明ACFs-S-nZVI/PMS体系的催化性能对反应温度的依赖性较小。此外,实际废水往往成分复杂,pH范围较广,因此催化剂的pH适应性是考察其实际应用潜力的重要指标,而由于PMS与催化剂的反应会使溶液pH发生变化[28],因此,本研究首先探究了初始pH在2、4、6、8和10时,反应溶液pH随时间的变化,结果如图4(b)插图所示。可见,在不同初始pH下,ACFs-S-nZVI/PMS体系pH在加入PMS后显著下降,当初始pH为2、4和6时,体系pH稳定在1.99、3.55、3.83左右,当初始pH分别为8和10时,体系pH缓慢上升至4.01和4.85,为弱酸性。TCP降解结果表明(图4(b)),当初始pH由2增加到10,ACFs-S-nZVI/PMS体系对浓度为10 μmol·L−1 TCP的去除率仍能达到98%左右,可见,即使PMS引入会使体系的初始pH迅速下降,但在后面的弱酸反应环境下ACFs-S-nZVI/PMS体系仍可实现对污染物的有效去除,说明ACFs-S-nZVI/PMS体系受pH的影响较小,适应于复杂环境下的污染物处理。这可能是因为硫化的nZVI相比于初始nZVI具有更高的环境适用性,可确保ACFs-S-nZVI在宽的pH范围内仍然具有较高的反应活性[29]。
催化剂用量对浓度为10 μmol·L−1 TCP的去除率的影响如图4(c)所示,当ACFs-S-nZVI用量由0.2 g·L−1增加到0.5 g·L−1时,TCP的去除率也随之提高。这一现象说明随着催化剂用量的增加,更多的催化活性位点被暴露,从而促进了PMS的活化和TCP的降解[30]。此外,当PMS浓度由0.25 mmol·L−1增加到0.5 mmol·L−1时,TCP(10 μmol·L−1)的去除速率也逐渐增加(图4(d)),然而,随着PMS浓度进一步提高至2 mmol·L−1时,TCP的去除速率反而有所减弱。这是因为体系中存在过量的PMS,其产生的大量ROS发生了自淬灭反应,导致PMS被无效消耗,从而抑制了TCP的降解[31]。
-
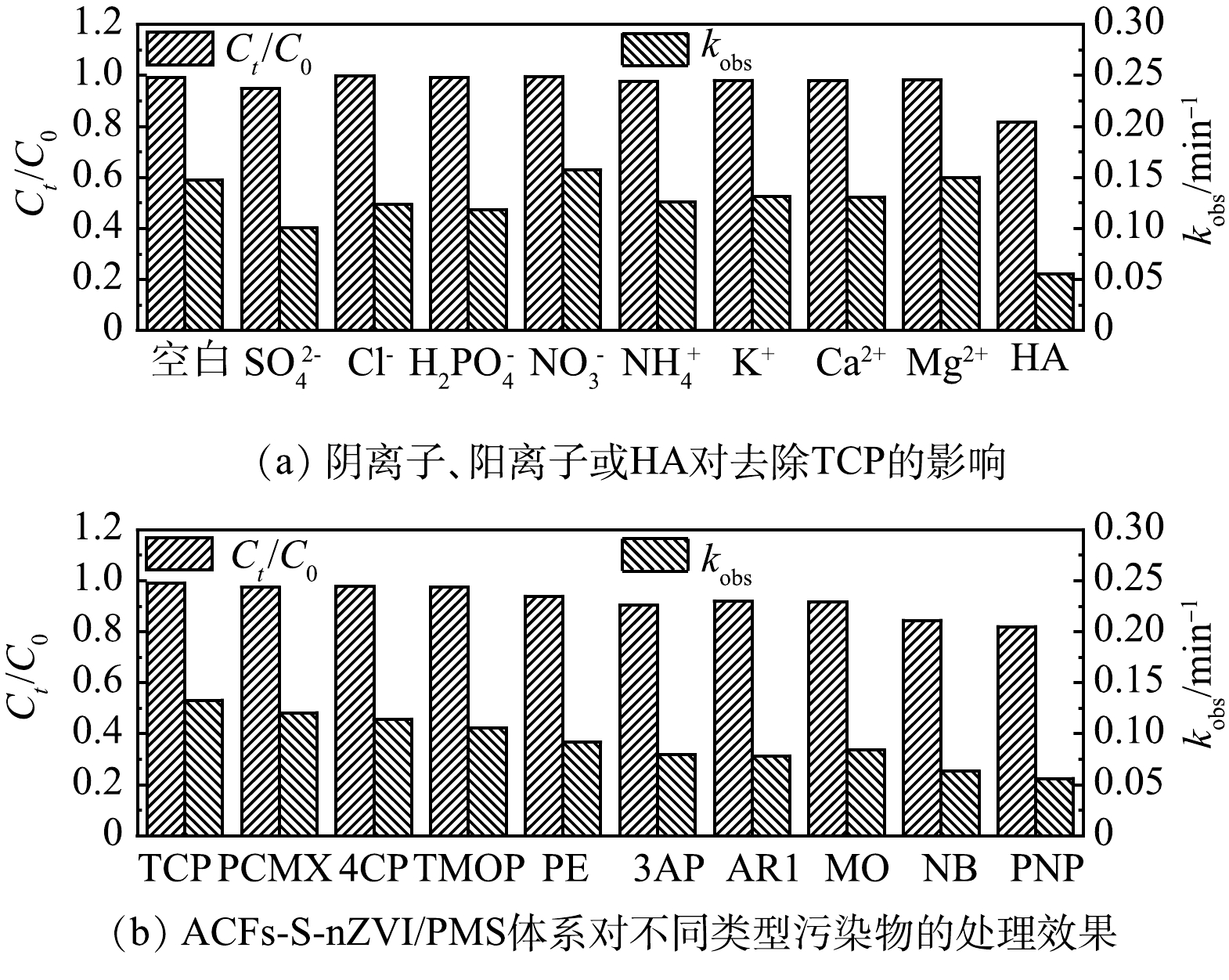
实际水体中往往会存在多种无机盐离子和一些天然有机物质,这些物质会对催化剂的性能产生负面影响[32]。因此,研究了几种常见的无机盐离子(SO42−、Cl−、H2PO4−、NO3−、NH4+、K+、Ca2+和Mg2+)和腐殖酸(humic acid,HA)对ACFs-S-nZVI/PMS体系去除浓度为10 μmol·L−1 TCP的影响。如图5(a)所示,ACFs-S-nZVI/PMS体系在加入离子后仍能去除97%以上的TCP。说明无机盐离子对该体系的影响较小。但加入HA后,TCP的去除率下降到81.66%,说明HA的存在抑制了TCP的去除。这是因为HA是一种典型的天然有机物质,其在催化反应中会与污染物形成竞争关系[33]。
除此之外,为了进一步探究ACFs-S-nZVI/PMS体系对不同有机污染物的普适性,选取了多种有机污染物进行去除[34-40]。如图5(b)所示,ACFs-S-nZVI/PMS体系对4-氯-3,5-二甲基苯酚(4-chloro-3,5-dimethylphenol,PCMX)、对氯苯酚(4-chlorophenol,4CP)、3-甲氧基苯酚(3-methoxyphenol,TMOP)、苯酚(phenol,PE)、3-氨基苯酚(3-aminophenol,3AP)、酸性红1(acid red 1,AR1)及甲基橙(methyl orange,MO)的去除率均能达到90%以上,而且对难降解的硝基苯(nitrobenzene,NB)和对硝基苯酚(p-nitrophenol,PNP)的去除率也可达到84.46%和82.07%。这表明ACFs-S-nZVI/PMS体系对不同有机污染物都具有较好的去除效果。
-
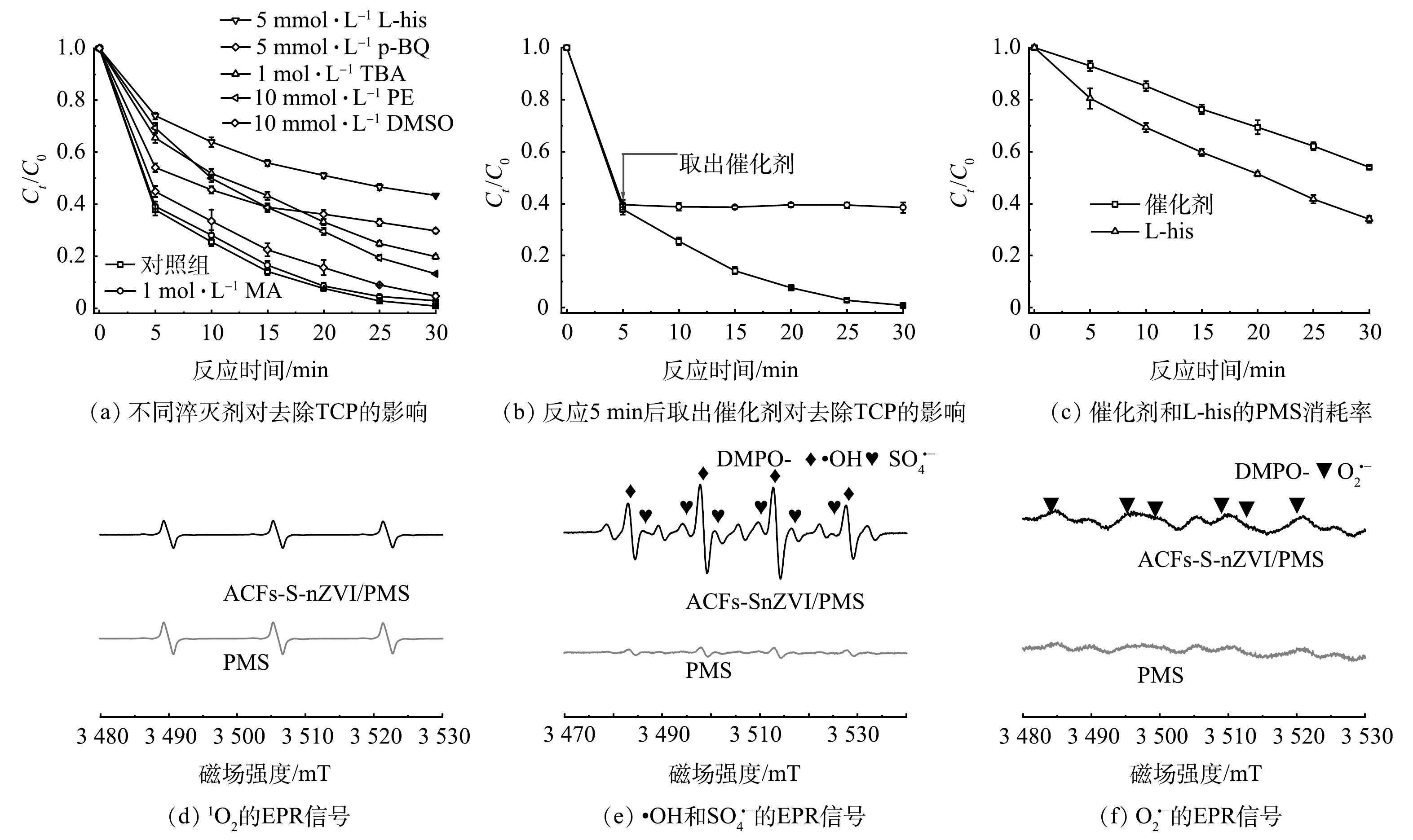
1)活性物种种类及贡献。为了探究ACFs-S-nZVI/PMS体系去除TCP(10 μmol·L−1)的机理,本研究通过淬灭实验考察了•OH、硫酸根自由基(SO4•−)、超氧自由基(O2•−)、单线态氧(1O2)和高价态铁(常见活性物种种类)在催化反应中的作用,结果如6(a)所示。首先,选择叔丁醇(tert-butanol,TBA)作为•OH的淬灭剂((k(TBA-•OH)=3.8~7.6×108 L·(mol·s)−1,远高于k(TBA-SO4•−)=4.0~9.1×105 L·(mol·s)−1)[27],发现TCP的去除率在添加过量TBA后降低了19%,说明•OH可能参与了ACFs-S-nZVI/PMS体系的催化反应。其次,甲醇(methanol,MA)一般作为•OH和SO4•−的淬灭剂(k(MA-•OH)=9.7×108 L·(mol·s)−1,k(MA-SO4•−)=1.0×107 L·(mol·s)−1)[41],但TCP的去除在加入过量MA后仅被轻微抑制,这可能是因为催化反应主要发生在催化剂表面,亲水的MA难以淬灭吸附在催化剂表面的自由基,而更具疏水性的TBA则会附着在催化剂表面,从而抑制了TCP的降解[42]。此外,PE结构中具有疏水性的苯环,所以PE容易聚集在材料表面,可用作非均相体系中表面自由基的淬灭剂[43]。当加入过量PE后,TCP的去除率被抑制了13%,这与TBA的抑制率相近,证明了表面自由基的存在。另外,在反应进行5 min时取出催化剂,然后于相同的反应条件下处理剩余溶液中的TCP,结果如图6(b)所示,继续反应25 min后,几乎没有TCP被去除,说明随着催化剂的取出,表面自由基消失,TCP的去除率被显著抑制,进而证明了表面自由基对ACFs-S-nZVI/PMS体系中TCP的去除具有重要作用。对苯醌(p-benzoquinones,p-BQ)通常作为O2•−的淬灭剂(k(p-BQ-O2•−)=1.9×109 L·(mol·s)−1)[44],TCP的去除率在加入过量p-BQ后被抑制了29%,说明O2•−可能在TCP去除中也起到了一定的作用。而L-组氨酸(L-histidine,L-his)常用于淬灭1O2(k(L-his-1O2)=2×109 L·(mol·s)−1)[44],加入过量L-his后,TCP的去除率被抑制了43%,说明1O2可能为去除TCP作出了贡献。随后,为了进一步验证体系中是否存在1O2,我们采用EPR以及捕获剂TEMP对其进行鉴定。如图6(d)所示,加入ACFs-S-nZVI催化剂之后,TEMP-1O2加合物的强度几乎没有变化,表明ACFs-S-nZVI/PMS体系中引起污染物降解的主要活性物种不是1O2。为了探究TCP去除率在添加L-his后被抑制的原因,分别用KSCN和KI显色法检测了L-his和催化剂消耗的PMS,结果如图6(c)所示,L-his消耗了溶液中66%的PMS,这远远大于催化剂自身消耗的PMS(46%),说明在ACFs-S-nZVI/PMS体系中引入L-his后,PMS的有效浓度大大降低,从而令TCP的降解受到了抑制,进一步证明了1O2不是体系中的主要ROS。有研究报道,在铁与过硫酸盐反应的过程中可能会有高价态铁的产生,而高价态铁活性物种具有氧化性可用于降解污染物[45],因此,我们选择二甲基亚砜(dimethylsulphoxide,DMSO)作为高价态铁淬灭剂,以此判断ACFs-S-nZVI/PMS体系中是否有高价态铁活性物种生成[46],结果如图6(a)所示。可见,在体系中加入过量DMSO后,仍然有95%以上的TCP被去除,说明ACFs-S-nZVI/PMS体系中引起污染物降解的主要活性物种不是高价铁。
除此之外,为了更加直观地验证ACFs-S-nZVI/PMS体系中是否存在•OH、SO4•−和O2•−,实验采用DMPO作为捕获剂,结果如图6(e)所示。ACFs-S-nZVI/PMS体系在水溶液中检测到了DMPO-•OH和DMPO-SO4•−加合物的强信号。证明了反应过程中确实有•OH、SO4•−的参与。此外,研究还将DMPO引入MA溶液以排除其它ROS的干扰,结果出现了特征是六重峰的DMPO-O2•−加合物信号(图6(f)),证实了ACFs-S-nZVI/PMS体系中O2•−的存在。综上,MA、TBA、p-BQ和L-his淬灭实验结合EPR结果表明,ACFs-S-nZVI/PMS体系中产生的ROS(•OH、SO4•−和O2•−)为去除TCP做出了重要贡献。
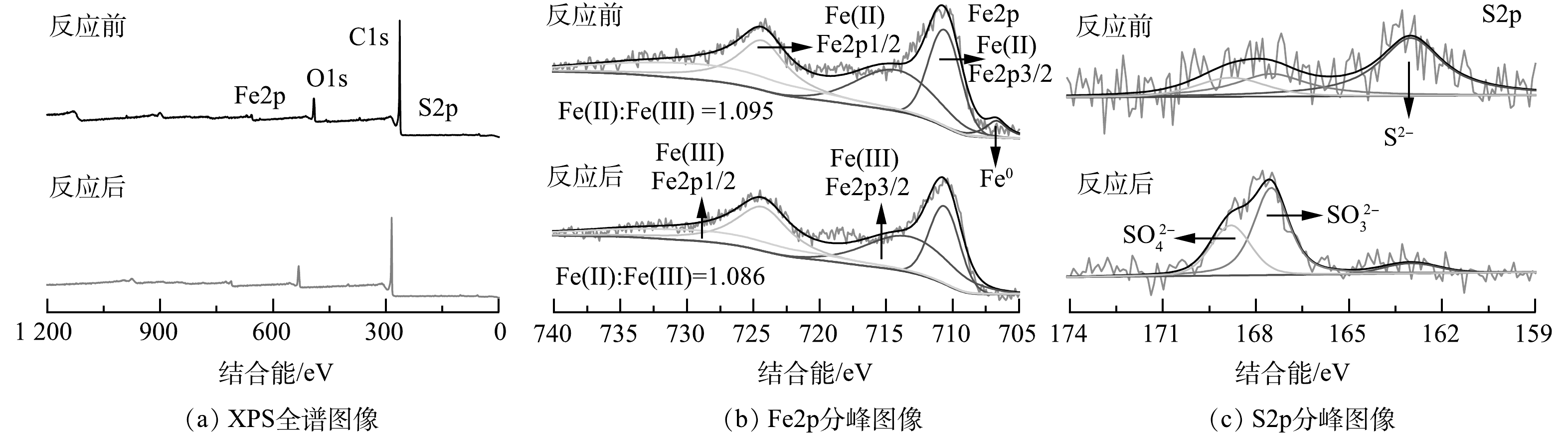
2)体系催化降解机理。为进一步分析ACFs-S-nZVI/PMS体系的催化机理,本文通过X射线光电子能谱(XPS)对ACFs-S-nZVI反应前后材料表面的元素组成、含量及价态变化进行了表征。XPS全谱图(图7(a))表明ACFs-S-nZVI中存在Fe、O、C和S元素,进一步证实了ACFs-S-nZVI催化剂的成功制备。一般而言,在金属负载型催化剂活化PMS的体系中,金属可以与PMS反应生成多种ROS,而硫元素的掺杂能促进体系内的电子转移[47]。因此,研究ACFs-S-nZVI反应前后Fe、S元素的化学变化是有必要的。其中,Fe 2p的光谱拟合如图7(b)所示,反应前在707.1 eV处的特征峰归属于Fe0,但Fe0含量较低,这可归因于制备过程中表面Fe0被氧化而生成铁氧化物或铁硫化物,反应后Fe0特征峰消失,说明随着反应的进行,Fe0作为电子供体被消耗,进而促进了Fe(Ⅲ)与Fe(Ⅱ)之间的循环(式(2))[48]。同时,在710.6、714.2、724.3和728.5 eV处分别出现了属于Fe(Ⅱ) Fe2p3/2、Fe(Ⅲ) Fe2p3/2、Fe(Ⅱ) Fe2p1/2和Fe(Ⅲ) Fe2p1/2的特征峰,经过反应之后,Fe(Ⅱ)和Fe(Ⅲ)的含量比从反应前的1.095略微下降至1.086,进一步证明了Fe0能促进Fe(Ⅲ)转化为Fe(Ⅱ)。而反应后Fe(Ⅱ)比例下降,可归因于Fe(Ⅱ)将电子传递至PMS,致使PMS被活化而产生•OH、SO4•−和O2•−,同时Fe(Ⅱ)被转化为Fe(Ⅲ)(式(3)、式(4)和式(5))[49]。此外,研究还对S2p光谱进行了拟合分析,结果如图7(c)所示,在163.2、167.9和169.9 eV处的特征峰分别归属于S2−、SO32−和SO42−。反应后,S2−的百分比明显减小,SO32−、SO42−的百分比显著增加,这一结果预示着催化剂在活化PMS的过程中,表面的S2−易失去电子而转化为SO32−、SO42−,进一步减缓了Fe的氧化(式(6))[50]。
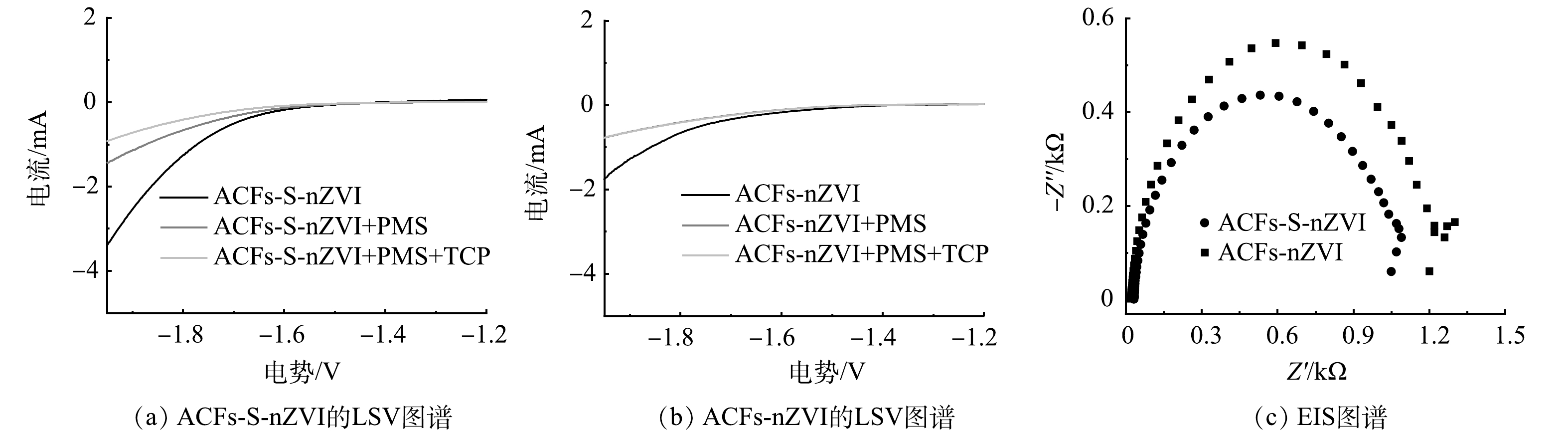
据报道,硫掺杂能提高nZVI电子转移的能力[51],为此本研究通过LSV和EIS对比了ACFs-S-nZVI与ACFs-nZVI的电子性能,并研究了电子传递在ACFs-S-nZVI/PMS体系中发挥的作用。LSV测试通过改变电极材料或电解液组分可获取不同催化条件下的响应电流值大小,进而能够分析反应体系的电子转移效应。如图8(a)~(b)所示,ACFs-S-nZVI的初始响应电流值高于ACFs-nZVI,说明ACFs-S-nZVI的电子传递性能更优。当向体系中加入PMS后,电流响应值明显降低,这一结果表明,ACFs-S-nZVI在活化PMS过程中可以作为电子供体,使得电子从ACFs-S-nZVI向PMS转移,进而活化PMS产生ROS,该过程与此前文献报道的结果一致[52]。当同时添加PMS和TCP时,ACFs-S-nZVI体系电流响应值的变化与ACFs-nZVI相比更为显著,说明ACFs-S-nZVI、PMS和TCP三者共存极大地促进了体系内的电子转移[42]。
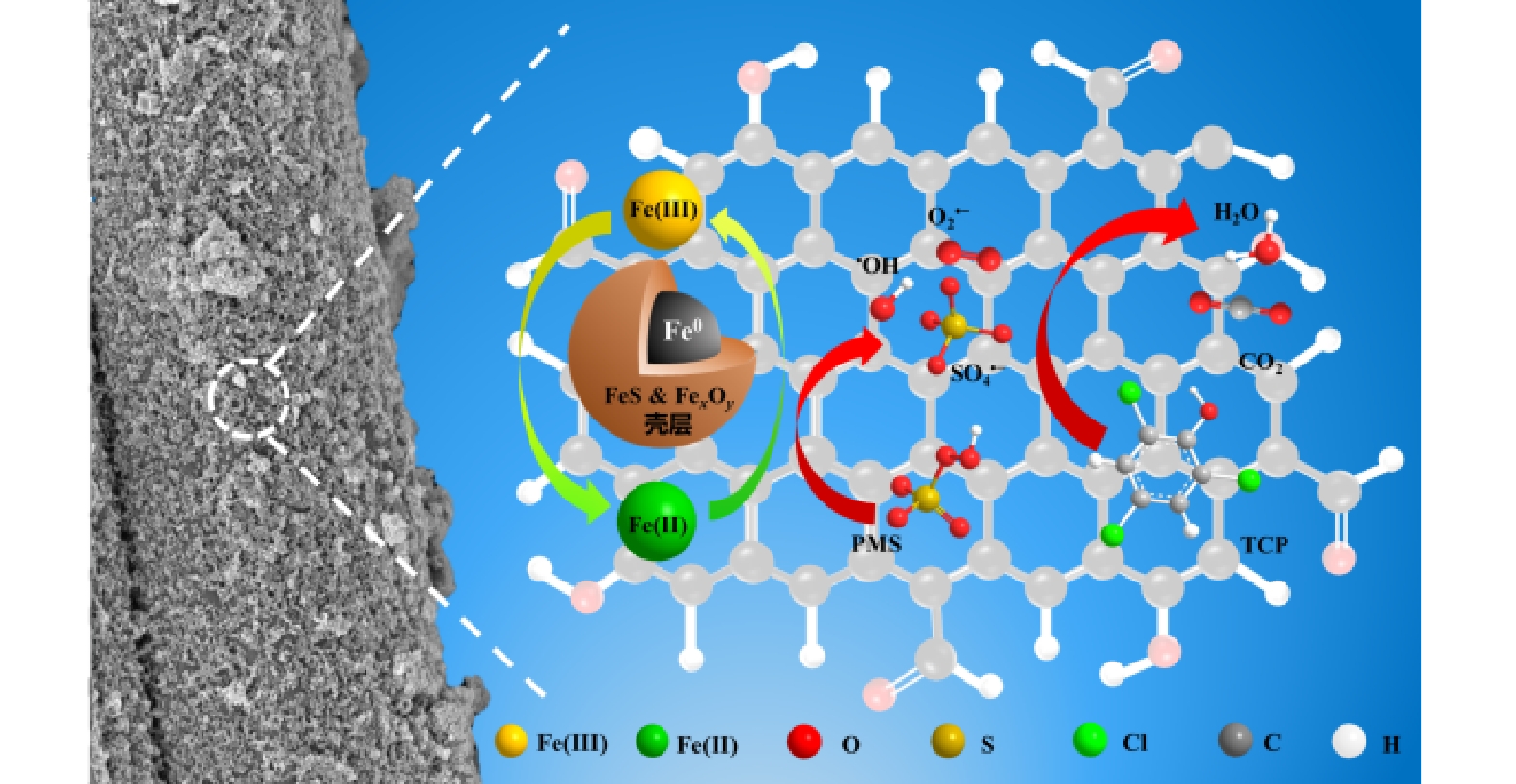
此外,根据Nyquist图中的圆弧直径能判断催化剂表面电子迁移阻力的大小[44]。如图8(c)所示,ACFs-S-nZVI样品的圆弧直径远小于ACFs-nZVI样品,说明ACFs-S-nZVI的表面电子迁移阻力更小,即电子传递速率更快,这与LSV测试结论一致,也为硫化能显著提高材料的电子转移能力提供了有力证据。基于以上研究,提出了如下可能的去除机制(图9):首先TCP被吸附在催化剂表面,然后催化剂表面电子转移至PMS,致使PMS被活化而产生ROS,TCP在ROS作用下脱氯氧化而转化为有机中间体,这些有机中间体再经过ROS的进一步分解而转化为H2O和CO2。
-
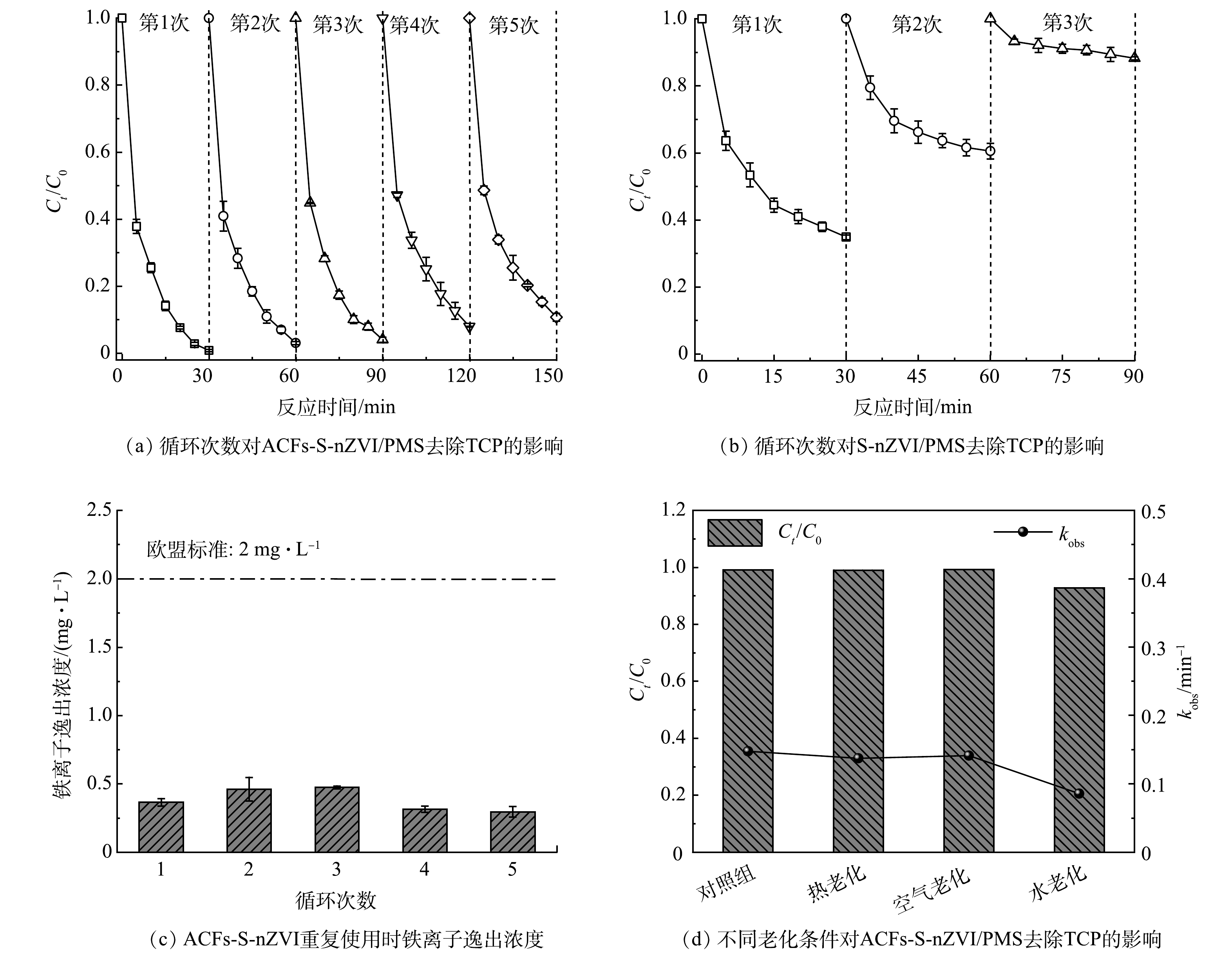
催化剂的重复使用性能是确定催化剂能否稳定应对环境修复中复杂问题的重要参考标准。因此,设计了如下循环实验来考察ACFs-S-nZVI材料的重复使用性:每完成1组浓度为10 μmol·L−1的TCP降解实验,即立刻取出催化剂(ACFs-S-nZVI采用镊子夹取法回收,S-nZVI采用磁分离技术回收),并用去离子水和无水乙醇交替洗涤,然后转移到新的TCP(10 μmol·L−1)中并重新加入PMS,无需额外操作,结果如图10(a)~(b)所示。可见,ACFs-S-nZVI/PMS体系对TCP的去除率从第1次循环的99.14%缓慢下降到第5次循环的89.22%,而S-nZVI/PMS体系经过5次循环后对TCP的去除率由65.13%显著下降到11.80%,说明ACFs-S-nZVI具有更优异的重复使用性。这是因为S-nZVI作为粉末状催化剂,在溶液中以高度分散的悬浮液形式存在,回收时损耗极大,处置不当还可能会引起二次污染,而ACFs-S-nZVI复合材料呈块状易分离回收,且以ACFs作为载体提高了S-nZVI的分散性和稳定性,进一步增强了ACFs-S-nZVI的重复使用性[53]。此外,结合图7(a)~(c)使用前后元素的变化对ACFs-S-nZVI循环后性能下降的原因进行分析,发现Fe、S元素发生了氧化,且使用后O元素百分比从6.57%提升至9.44%,说明催化剂结构及表面活性位点可能被TCP的氧化产物所覆盖,导致ACFs-SnZVI/PMS体系对TCP的去除率下降[54]。此外,为了探究ACFs-S-nZVI在重复使用后的稳定性情况,每完成1组循环实验,及时测量剩余溶液中的铁离子浓度,结果如图10(c)所示,ACFs-S-nZVI 5次循环逸出的铁离子都小于0.5 mg·L−1,远低于欧盟标准(2 mg·L−1),说明催化剂在循环中具有很高的稳定性,进一步证明ACFs载体对S-nZVI有很强的固定化作用[55]。
一般而言,催化剂在实际使用过程中会受到氧化对其结构和反应活性产生的负面影响[56]。因此,探究ACFs-S-nZVI材料的抗氧化性是有必要的,同时,考虑到实际使用时复杂的环境,分别采用80 ℃热水、空气、去离子水对催化剂进行一定时间的老化处理,而后将其用于浓度为10 μmol·L−1的TCP去除实验,结果如图10(d)所示,经老化处理后的ACFs-S-nZVI仍然可以去除92%以上的TCP,说明ACFs-S-nZVI具有优异的抗氧化性,这是因为nZVI经负载和硫化改性后,减少了与O2和H2O的接触,进而延长了ACFs-S-nZVI的使用寿命[57]。因此,可以预见ACFs-S-nZVI在实际应用中也具有较强的催化活性和适应性[58]。
为进一步考察该体系在实际废水中对TCP的处理效果,参考实际TCP废水中的物质组分及含量配置了模拟TCP废水。根据中国《废水综合排放标准》(GB 8798―1996),排入废水处理厂的芳烃污染物应符合3类标准,质量浓度范围为1~5 mg·L−1。此外,酚类合成废水中的污染物初始质量浓度一般超过2 000 mg·L−1,该种含酚废水经预处理后可回收大部分的酚类物质,同时废水中含酚质量浓度也降至10 mg·L−1。因此本研究将模拟废水中TCP的质量浓度设定为10 mg·L−1。此外,废水中还包含一些常见的阴阳离子(SO42−、Cl−、H2PO4−、NO3−、NH4+、K+、Ca2+、Mg2+)和HA,其中,除Cl−离子浓度设置为50 mmol·L−1外,其它离子浓度为10 mmol·L−1,HA质量浓度为20 mg·L−1。结果如图11所示,ACFs-S-nZVI/PMS体系对模拟的实际废水也具备良好的去除效果,在30 min内可以去除78.69%质量浓度为10 mg·L−1的TCP,进一步验证了该体系具备良好的实际应用潜力。
-
1)硫掺杂和以ACFs作为载体可以显著提升nZVI的催化性能,改性后的催化剂颗粒不仅变得更加分散和均匀,而且直径得到显著减小。
2)当反应条件设置为S/Fe比0.3、温度25 ℃、pH=6、催化剂投加量0.5 g·L−1、PMS投加量0.5 mmol·L−1时,ACFs-S-nZVI/PMS体系在30 min内即可去除99.14%的TCP(10 μmol·L−1),明显优于改性前的催化体系。
3) •OH、SO4•−和O2•−在ACFs-S-nZVI/PMS体系去除TCP中发挥着重要作用;硫化能显著提高ACFs-S-nZVI的电子传递能力。
4) ACFs-S-nZVI/PMS体系在去除TCP(10 μmol·L−1)时表现出了优异的酸碱耐受性(pH=2~10)和抗离子干扰性;即使经过5次循环或不同的老化处理后,也仍能达到90%左右的TCP去除率;在处理模拟TCP(10 mg·L−1)废水时,效果同样较好,表明ACFs-S-nZVI/PMS体系在废水的实际处理中具有广阔的应用前景。
活性炭纤维负载硫化纳米零价铁用于活化PMS降解三氯苯酚
Degradation of trichlorophenol by activated carbon fibers-supported S-nZVI activating PMS
-
摘要: 针对纳米零价铁(nanoscale zero-valent iron,nZVI)在反应过程中易团聚、易钝化和难回收等问题,采用比表面积大、化学稳定性好的活性炭纤维(activated carbon fibers,ACFs)作为载体,以连二亚硫酸钠(Na2S2O4)作为硫化试剂,制备活性炭纤维负载硫化纳米零价铁复合材料(activated carbon fibers supported sulfidized nanoscale zero-valent iron,ACFs-S-nZVI),并考察了复合材料活化过一硫酸盐(peroxymonosulfate,PMS)降解三氯苯酚(trichlorophenol,TCP)的催化性能、反应机理及重复使用性能。材料表征结果表明,硫化不仅可以缓解nZVI的氧化,还可以加快nZVI的电子传输速率,而引入ACFs载体则极大地提高了硫化纳米零价铁(sulfidized nanoscale zero-valent iron,S-nZVI)的分散性和稳定性。通过条件探究实验发现,当反应条件设置为S/Fe比0.3、温度25 ℃、pH=6、催化剂投加量0.5 g·L−1、PMS投加量0.5 mmol·L−1时,ACFs-S-nZVI/PMS体系表现出最佳的催化性能,在30 min内即可去除99.14% 10 μmol·L−1的TCP,显著优于未改性的样品。此外,该体系具有较宽的pH(2~10)适应范围、优异的抗离子干扰能力以及出色的重复使用性能。电子顺磁共振波谱(EPR)和自由基淬灭实验结果表明,羟基自由基(•OH)、硫酸根自由基(SO4•−)和超氧自由基(O2•−)参与了ACFs-S-nZVI/PMS体系的催化反应。综上可知,ACFs-S-nZVI具有催化性能优异、稳定性高、环境适应性强等优点,在催化降解有机污染物方面表现出较好的应用前景。Abstract: In order to cope with the problems of severe agglomeration, easy passivation and difficult recovery of nanoscale zero-valent iron (nZVI) during reaction process, the activated carbon fibers (ACFs) with large specific surface area and excellent chemical stability were taken as carriers, and sodium dithionite (Na2S2O4) was taken as the sulfidation reagent to successfully fabricate activated carbon fibers-supported sulfidized nanoscale zero-valent iron composite (ACFs-S-nZVI). Subsequently, the catalytic performance, reaction mechanism and recycling performance of this composite catalyst on trichlorophenol (TCP) degradation by activating peroxymonosulfate (PMS) were further investigated. The results of material characterization showed that the deposited ferrous sulfide (FeS) during the vulcanization process could not only mitigate the oxidation of nZVI, but also accelerate the electron transfer rate of nZVI, and the introduction of ACFs carrier substantially improved the dispersion and stability of sulfidized nanoscale zero-valent iron (S-nZVI). Through operational condition experiments, it was found that the ACFs-S-nZVI/PMS system exhibited the best catalytic performance when the reaction conditions were set to S/Fe ratio of 0.3, 25 ℃, pH of 6, catalyst dosage of 0.5 g·L−1 and PMS dosage of 0.5 mmol·L−1, and 99.14% of TCP with a concentration of 10 μmol·L−1 could be degraded within 30 min, which was significantly better than that of the unmodified sample. In addition, the system had a wide pH adaptation range (pH=2~10), an excellent anti-ion interference ability and an outstanding recycling performance. The electron paramagnetic resonance (EPR) spectroscopy and radical quenching experiments showed that hydroxyl radical (•OH), sulfate radical (SO4•−) and superoxide radical (O2•−) participated in the catalytic reaction of ACFs-S-nZVI/PMS system. In summary, ACFs-S-nZVI has the advantages of excellent catalytic performance, high stability, and strong environmental adaptability. Therefore, it shows great application prospects in catalytic degradation of organic pollutants.
-

-
-
[1] 王双, 杜倩, 谭莉, 等. AgBr-WO3/GO载流子转移增强及其可见光降解2, 4, 6-三氯苯酚[J]. 功能材料, 2022, 53(2): 2130-2134. doi: 10.3969/j.issn.1001-9731.2022.02.019 [2] 张万辉. 零价铁对2, 4-二氯酚的还原脱氯研究[J]. 广东化工, 2014, 41(13): 47-48. doi: 10.3969/j.issn.1007-1865.2014.13.023 [3] LIANG J L, ZHOU Y. Iron-based advanced oxidation processes for enhancing sludge dewaterability: State of the art, challenges, and sludge reuse[J]. Water Research, 2022, 218: 118499. doi: 10.1016/j.watres.2022.118499 [4] AHMED Y, ZHONG J X, YUAN Z G, et al. Roles of reactive oxygen species in antibiotic resistant bacteria inactivation and micropollutant degradation in Fenton and photo-Fenton processes[J]. Journal of Hazardous Materials, 2022, 430: 128408. doi: 10.1016/j.jhazmat.2022.128408 [5] SUN Y M, ZHOU P, ZHANG P, et al. New insight into carbon materials enhanced Fenton oxidation: A strategy for green iron (III) /iron (II) cycles[J]. Chemical Engineering Journal, 2022, 450: 138423. doi: 10.1016/j.cej.2022.138423 [6] MEYERSTEIN D. Re-examining Fenton and Fenton-like reactions[J]. Nature Reviews Chemistry, 2021, 5(9): 595-597. doi: 10.1038/s41570-021-00310-4 [7] KAVITHA V, PALANIVELU K. Degradation of phenol and trichlorophenol by heterogeneous photo-Fenton process using granular ferric hydroxide®: Comparison with homogeneous system[J]. International Journal of Environmental Science and Technology, 2016, 13(3): 927-936. doi: 10.1007/s13762-015-0922-y [8] WANG N N, ZHENG T, ZHANG G S, et al. A review on Fenton-like processes for organic wastewater treatment[J]. Journal of Environmental Chemical Engineering, 2016, 4(1): 762-787. doi: 10.1016/j.jece.2015.12.016 [9] FAROOQI Z H, BEGUM R, NASEEM K, et al. Zero valent iron nanoparticles as sustainable nanocatalysts for reduction reactions[J]. Catalysis Reviews, 2022, 64(2): 286-355. doi: 10.1080/01614940.2020.1807797 [10] LI Y R, ZHAO H-P, ZHU L Z. Remediation of soil contaminated with organic compounds by nanoscale zero-valent iron: a review[J]. Science of the Total Environment, 2021, 760: 143413. doi: 10.1016/j.scitotenv.2020.143413 [11] XU B D, LI D C, QIAN T T, et al. Boosting the activity and environmental stability of nanoscale zero-valent iron by montmorillonite supporting and sulfidation treatment[J]. Chemical Engineering Journal, 2020, 387: 124063. doi: 10.1016/j.cej.2020.124063 [12] LI J X, ZHANG X Y, SUN Y K, et al. Advances in sulfidation of zerovalent iron for water decontamination[J]. Environmental Science & Technology, 2017, 51(23): 13533-13544. [13] MO Y L, XU J, ZHU L Z. Molecular structure and sulfur content affect reductive dechlorination of chlorinated ethenes by sulfidized nanoscale zerovalent iron[J]. Environmental Science & Technology, 2022, 56(9): 5808-5819. [14] 郭雅妮, 强雪妮, 李海红, 等. 不同预处理方法对活性炭纤维结构和吸附性能的影响[J]. 环境工程学报, 2016, 10(5): 2227-2232. doi: 10.12030/j.cjee.201412128 [15] ZHANG C C, TIAN H F, WANG Z X, et al. Degradation of PAHs in soil by activated persulfate system with activated carbon supported iron-based bimetal[J]. Science of the Total Environment, 2023, 866: 161323. doi: 10.1016/j.scitotenv.2022.161323 [16] DONG H R, DENG J M, XIE Y K, et al. Stabilization of nanoscale zero-valent iron (nZVI) with modified biochar for Cr (VI) removal from aqueous solution[J]. Journal of Hazardous Materials, 2017, 332: 79-86. doi: 10.1016/j.jhazmat.2017.03.002 [17] 苏冰琴, 温宇涛, 林昱廷, 等. 改性活性炭纤维活化过硫酸盐深度处理焦化废水及降解吡啶的反应机制研究[J]. 中国环境科学, 2023, 43(2): 576-591. doi: 10.3969/j.issn.1000-6923.2023.02.010 [18] 甄建政, 聂士松, 潘世元, 等. 多维度碳基负载金属催化剂活化PMS降解水中污染物的研究进展[J]. 化工进展, 2022, 41(4): 1858-1872. doi: 10.16085/j.issn.1000-6613.2021-0738 [19] TAN W T, RUAN Y, DIAO Z H, et al. Removal of levofloxacin through adsorption and peroxymonosulfate activation using carbothermal reduction synthesized nZVI/carbon fiber[J]. Chemosphere, 2021, 280: 130626. doi: 10.1016/j.chemosphere.2021.130626 [20] 杨思明, 刘爱荣, 刘静, 等. 硫化纳米零价铁研究进展: 合成、性质及环境应用[J]. 化学学报, 2022, 80(11): 1536-1554. [21] KONG A Q, LIU M H, ZHANG H J, et al. Highly selective electrocatalytic hydrogenation of benzoic acid over Pt/C catalyst supported on carbon fiber[J]. Chemical Engineering Journal, 2022, 445: 136719. doi: 10.1016/j.cej.2022.136719 [22] SUN J A, WANG L X, WANG Y G, et al. Activation of peroxymonosulfate by MgCoAl layered double hydroxide: Potential enhancement effects of catalyst morphology and coexisting anions[J]. Chemosphere, 2022, 286: 131640. doi: 10.1016/j.chemosphere.2021.131640 [23] XU J, AVELLAN A, LI H, et al. Sulfur loading and speciation control the hydrophobicity, electron transfer, reactivity, and selectivity of sulfidized nanoscale zerovalent iron[J]. Advanced Materials, 2020, 32(17): 1906910. doi: 10.1002/adma.201906910 [24] XU J, AVELLAN A, LI H, et al. Iron and sulfur precursors affect crystalline structure, speciation, and reactivity of sulfidized nanoscale zerovalent iron[J]. Environmental Science & Technology, 2020, 54(20): 13294-13303. [25] XU Y N, WU Y T, LIU Y F, et al. Covering extracellular polymeric substances to enhance the reactivity of sulfidated nanoscale zerovalent iron toward Cr (VI) removal[J]. Chemical Engineering Journal, 2022, 448: 137610. doi: 10.1016/j.cej.2022.137610 [26] DUAN X G, SU C, ZHOU L, et al. Surface controlled generation of reactive radicals from persulfate by carbocatalysis on nanodiamonds[J]. Applied Catalysis B: Environmental, 2016, 194: 7-15. doi: 10.1016/j.apcatb.2016.04.043 [27] LI J W, ZOU J, ZHANG S Y, et al. Sodium tetraborate simultaneously enhances the degradation of acetaminophen and reduces the formation potential of chlorinated by-products with heat-activated peroxymonosulfate oxidation[J]. Water Research, 2022, 224: 119095. doi: 10.1016/j.watres.2022.119095 [28] 李广英, 杜敏洁, 谈成英, 等. 锰铁氧体活化PMS降解双酚A的过程机制[J]. 环境工程学报, 2021, 15(9): 2952-2962. [29] DU J K, BAO J G, LU C H, et al. Reductive sequestration of chromate by hierarchical FeS@Fe (0) particles[J]. Water Research, 2016, 102: 73-81. doi: 10.1016/j.watres.2016.06.009 [30] 姚梦东, 岳俊杰, 徐雪婧, 等. 球磨硫化零价铁活化过硫酸盐降解水体中有机氯农药[J]. 环境工程学报, 2021, 15(8): 2563-2575. doi: 10.12030/j.cjee.202103052 [31] 李鑫, 尹华, 罗昊昱, 等. 磁性生物炭负载α-MnO2活化过一硫酸盐降解2, 2′, 4, 4′-四溴联苯醚[J]. 环境科学, 2021, 42(10): 4798-4806. [32] LI D P, FENG Z Q, ZHOU B H, et al. Impact of water matrices on oxidation effects and mechanisms of pharmaceuticals by ultraviolet-based advanced oxidation technologies: A review[J]. Science of the Total Environment, 2022, 844: 157162. doi: 10.1016/j.scitotenv.2022.157162 [33] ZHEN J Z, NIE S S, SUN J H, et al. Fe3O4 nanoparticles encapsulated in boron nitride support via N-doped carbon layer as a peroxymonosulfate activator for pollutant degradation: Important role of metal boosted C–N sites[J]. Journal of Environmental Management, 2022, 311: 114859. doi: 10.1016/j.jenvman.2022.114859 [34] 杨佩汶, 林毅, 林华, 等. 不同构型人工湿地-微生物燃料电池对废水中对氯苯酚的净化效果及产电性能的影响[J]. 环境工程学报, 2023, 17(2): 507-516. doi: 10.12030/j.cjee.202210035 [35] 魏博. 大气和造纸废水中甲氧基苯酚类污染物去除机制的理论研究[D]. 济南: 山东大学, 2021. [36] DE FARIAS M B, PREDIGER P, VIEIRA M G A. Conventional and green-synthesized nanomaterials applied for the adsorption and/or degradation of phenol: A recent overview[J]. Journal of Cleaner Production, 2022, 367: 132980. doi: 10.1016/j.jclepro.2022.132980 [37] FANG C, HAO Z X, WANG Y L, et al. Carbon nanotube as a nanoreactor for efficient degradation of 3-aminophenol over CoOx/CNT catalyst[J]. Journal of Cleaner Production, 2023, 405: 136912. doi: 10.1016/j.jclepro.2023.136912 [38] 窦欣, 田乔鹏, 王琦, 等. Ganoderma sp. SYBC L48漆酶酶学性质及其对酸性红1的脱色性能[J]. 环境工程学报, 2019, 13(4): 856-864. doi: 10.12030/j.cjee.201809095 [39] 李新欣. 稻壳生物炭吸附水中对硝基苯酚和硝基苯的研究[D]. 南京: 南京信息工程大学, 2023. [40] CHEN Q, MA C R, DUAN W Y, et al. Coupling adsorption and degradation in p-nitrophenol removal by biochars[J]. Journal of Cleaner Production, 2020, 271: 122550. doi: 10.1016/j.jclepro.2020.122550 [41] TANG N N, QIAN C B, ZHANG C W, et al. Isolation anchoring strategy for fabricating high-loading uniformly dispersed iron-based catalysts toward selective removal of phenolic compounds[J]. Separation and Purification Technology, 2023, 326: 124789. doi: 10.1016/j.seppur.2023.124789 [42] CHEN L K, HUANG Y F, ZHOU M L, et al. Nitrogen-doped porous carbon encapsulating iron nanoparticles for enhanced sulfathiazole removal via peroxymonosulfate activation[J]. Chemosphere, 2020, 250: 126300. doi: 10.1016/j.chemosphere.2020.126300 [43] LIU X R, LIU Y, QIN H H, et al. Selective removal of phenolic compounds by peroxydisulfate activation: Inherent role of hydrophobicity and interface ROS[J]. Environmental Science & Technology, 2022, 56(4): 2665-2676. [44] WANG M X, WANG Y G, SUN J H, et al. Layered double hydroxide/carbonitride heterostructure with potent combination for highly efficient peroxymonosulfate activation[J]. Chemosphere, 2023, 313: 137394. doi: 10.1016/j.chemosphere.2022.137394 [45] DONG H Y, LI Y, WANG S C, et al. Both Fe (IV) and radicals are active oxidants in the Fe (II) /peroxydisulfate process[J]. Environmental Science & Technology Letters, 2020, 7(3): 219-224. [46] 胡彩萍, 锁进然, 丁冠涛, 等. 草酸强化天然铁矿石异相光助Fenton催化降解萘酚[EB/OL]. [2023-07-06]中国环境科学. DOI: 10.19674/j.cnki.issn1000-6923.20230508.001. [47] WU L B, LIN Q T, FU H Y, et al. Role of sulfide-modified nanoscale zero-valent iron on carbon nanotubes in nonradical activation of peroxydisulfate[J]. Journal of Hazardous Materials, 2022, 422: 126949. doi: 10.1016/j.jhazmat.2021.126949 [48] GAO F L, AHMAD S, TANG J C, et al. Enhanced nitrobenzene removal in soil by biochar supported sulfidated nano zerovalent iron: Solubilization effect and mechanism[J]. Science of the Total Environment, 2022, 826: 153960. doi: 10.1016/j.scitotenv.2022.153960 [49] LING C, WU S, HAN J A, et al. Sulfide-modified zero-valent iron activated periodate for sulfadiazine removal: Performance and dominant routine of reactive species production[J]. Water Research, 2022, 220: 118676. doi: 10.1016/j.watres.2022.118676 [50] JIANG Q, JIANG S M, LI H, et al. A stable biochar supported S-nZVI to activate persulfate for effective dichlorination of atrazine[J]. Chemical Engineering Journal, 2022, 431: 133937. doi: 10.1016/j.cej.2021.133937 [51] ZHANG C, LU J, WU J. One-step green preparation of magnetic seaweed biochar/sulfidated Fe0 composite with strengthen adsorptive removal of tetrabromobisphenol A through in situ reduction[J]. Bioresource Technology, 2020, 307: 123170. doi: 10.1016/j.biortech.2020.123170 [52] MI X Y, WANG P F, XU S Z, et al. Almost 100% peroxymonosulfate conversion to singlet oxygen on single‐atom CoN2+2 sites[J]. Angewandte Chemie International Edition, 2021, 60(9): 4588-4593. doi: 10.1002/anie.202014472 [53] XU Y, GUO Q, LI Y, et al. Stabilization of nano zero-valent iron by electrospun composite mat with good catalysis and recyclability[J]. Journal of Cleaner Production, 2022, 363: 132459. doi: 10.1016/j.jclepro.2022.132459 [54] QIN X D, LI Z K, ZHU Z W, et al. Mechanism and kinetics of treatment of acid orange II by aged Fe-Si-B metallic glass powders[J]. Journal of Materials Science & Technology, 2017, 33(10): 1147-1152. [55] WANG J H, DUAN H Y, WANG M X, et al. Construction of durable superhydrophilic activated carbon fibers based material for highly-efficient oil/water separation and aqueous contaminants degradation[J]. Environmental Research, 2021, 207: 112212. [56] DENG J M, CHEN T, ARBID Y, et al. Aging and reactivity assessment of nanoscale zerovalent iron in groundwater systems[J]. Water Research, 2022, 229: 119472. [57] XU J, WANG Y, WENG C, et al. Reactivity, selectivity, and long-term performance of sulfidized nanoscale zerovalent iron with different properties[J]. Environmental Science & Technology, 2019, 53(10): 5936-5945. [58] 陈砚田, 郄晗彤, 张胤杰, 等. 还原氧化石墨烯负载零价铁的合成及对TNT废水处理[J]. 高等学校化学学报, 2020, 41(8): 1836-1842. doi: 10.7503/cjcu20200198 -



 下载:
下载: