-
挥发性有机污染物(volatile organic compounds, VOCs)是生成光化学烟雾、PM2.5的重要前驱体,其种类繁多,部分属于有毒有害物质,会对自然环境和人身健康造成严重危害[1–4]。低温等离子体技术具有常温常压下能产生高能电子和活性物质、处理浓度范围广、对污染物无选择性及操作简单等优点,被广泛应用于对VOCs的处理[5–8]。
低温等离子体降解VOCs是利用放电产生的高能电子、自由基、臭氧等活性粒子,与VOCs分子碰撞后诱导激发其电离及解离,从而实现对VOCs的高效降解。常见低温等离子体反应器主要有电晕放电和介质阻挡放电(dielectric barrier discharge, DBD)两种。传统电晕放电装置常采用产生非均匀电场 (线-筒[9] 、线-板[10]或针-板[11]) 的电极结构。DBD反应器除采用非均匀电场电极的结构外,还可使用产生均匀电场 (板-板[12]、同轴圆筒[13]) 的电极结构。使用传统线-筒反应器处理VOCs的过程中,气体降解产生副产物气溶胶会粘附在线电极表面使放电间距减小,导致火花放电、甚至弧光放电,引起放电等离子体状态的改变,影响降解效果的稳定性,甚至可能导致反应器燃爆。在实际应用中,气溶胶沉积在电极表面会降低设备使用寿命,需要定时清洗或更换电极,造成运行和维护成本的增加。YAO等[14]利用线-板电极降解甲苯时发现,在线电极及板电极周围都会产生黄褐色气溶胶,并且随着时间的增加,气溶胶沉积量增加,颜色变深。ZHANG等[15]利用线-筒式反应器降解苯乙烯时发现随着处理时间和污染物浓度的增加,气溶胶等油类副产物容易沉积在反应器壁和线电极表面,这些沉积物影响了DBD运行状态及其对VOCs的降解性能。KARATUM等[16]观察到使用线-筒式反应器处理VOCs时,线电极表面及出口管呈现黄褐色,同时反应器的出口处积累了大量的深褐色小颗粒,且随着处理时间的增加,沉积物会造成反应器的堵塞。李杰等[17]采用V型收尘极板将电除尘器的粉尘收集区与放电区分开,解决了电除尘器收集高比电阻粉尘反电晕放电问题;有研究者改进了低温等离子体翘片-杆DBD电极结构,将翘片的凹槽用于沉积气溶胶副产物,翘片的刃沿用于产生放电,使DBD在实验时间内对VOCs的处理性能基本稳定[18–21]。但是由于DBD电极结构气隙间距小,会造成处理大流量VOCs污染气体时阻力大的问题。
本课题组将翘片-筒式电晕流光放电等离子体电极结构的放电区域(翘片尖端-筒)与副产物粘附区域(连接翘片的轴线)分开,研究翘片-筒式脉冲放电特性,优化翘片的布置方法,探究反应器对二氯甲烷降解效果及能量效率,以期获得工艺参数最优值,进而考察反应器运行的稳定性及气溶胶沉积情况,从而为低温等离子体处理VOCs废气的性能提升提供参考。
-
翘片-筒式脉冲电晕流光放电等离子体实验装置如图1所示。二氯甲烷标气、氮气、氧气由质量流量控制器(GT130D)控制进入混气瓶中,充分混合后稀释为实验模拟废气。在废气的组分稳定后进入等离子体反应器进行降解。等离子体反应器内部为翘片-筒结构,不锈钢圆筒厚度为0.2 cm,内径为9.8 cm;翘片结构由不锈钢螺纹杆(Φ0.8 cm)和3种不同直径的不锈钢翘片(Φ7 cm、Φ6 cm和Φ5 cm)组成,翘片等距排列在不锈钢螺纹杆上,与筒壁之间的距离(即放电间距B)分别为1.4 cm、1.9 cm和2.4 cm。放电有效长度(L)随着翘片的数量与间距的改变而变化。使用纳秒脉冲电源供电,利用示波器(Tektronix MDO3014)连接高压探头(Tektronix P6015A)和电流探头(Tektronix P6021A)测量放电过程中电流、电压及频率等参数。降解后的废气从反应器出口排出,分别进入气相色谱仪(Agilent 7890A)、傅里叶红外烟气分析仪(DX4000)以及气相-质谱联用仪(Agilent 7890B-5977B),以检测降解后的废气中二氯甲烷浓度以及中间产物的种类。利用便携式气体检测仪(TY2000-B)检测排放尾气中的HCl。
-
纳秒脉冲电源输入等离子体反应器的单个脉冲能量(W,mJ)由脉冲电压电流波形图积分计算而来,输入功率(P,W)为脉冲电源输入等离子体反应器的功率,计算公式见式(1)~(2)。
定义单位等离子体体积所注入的能量为能量体积密度EVD (energy volume density,mJ·L−1) ,计算公式见式(3)。
能量输入密度SIE(Specific input energy,J·L−1)是指单位体积二氯甲烷得到的能量,计算公式见式(4)。
二氯甲烷降解效率(η)为已降解污染物浓度与反应器进口浓度值的比值,计算公式见式(5)。
能量效率EY(Energy yield,g·kW h−1)指单位能量下去除二氯甲烷的质量,计算公式见式(6)。
式中:t为单个脉冲持续时间,s;
$ \text{U}\text{(}\text{t}\text{)} $ 和$ \text{I}\text{(}\text{t}\text{)} $ 分别为瞬时脉冲峰值电压和瞬时脉冲电流,kV和A;f为电源频率,HZ;V为等离子体放电区域体积,L;Q为气体流量,L·min−1;$ {\text{C}}_{\text{in}} $ 和$ {\text{C}}_{\text{out}} $ 分别为气体进口浓度值和出口浓度值,mg·m−3;M为二氯甲烷的摩尔质量,84.93 g·mol−1。 -
等离子体反应器放电功率及能量体积密度与其结构有密切联系。为最大限度将电能量传输到反应器当中,通过改变相邻翘片间距(A)与放电间距(B)来检测放电特性并优化电极配置。实验固定翘片数量为6片,调整相邻翘片间距,并以A/B的比值为变量来测试其对功率和能量体积密度的影响(图2)。3种放电间距下,随着峰值电压的增加,等离子体反应器的注入功率和能量体积密度均增加。在放电间距为2.4 cm时 (图2 (c) ) ,功率随着A/B值的增加而增大。当A/B值为1~1.5,功率增加缓慢基本保持平衡在1 W。随着A/B值的增加,能量体积密度呈现逐渐下降趋势。当A/B<1时,反应体系内虽能量体积密度较高,但注入功率相对较低,最小值为0.9 W;与之相反,当A/B>1时,反应体系内注入功率基本平衡,但能量体积密度较少,最低仅有16.2 mJ·L−1。当A/B=1时,反应器可兼顾功率和能量体积密度2个参数。此时,功率和能量体积密度在37 kV下的最大值分别为0.95 W和23.6 mJ·L−1。
当翘片间距较小时,相邻翘片之间会相互影响产生静电屏蔽,导致单次脉冲能量减少,进而使注入等离子体反应器的功率降低。相邻翘片间距的增加会造成放电区域长度与体积的增加,但由于注入反应器中的能量变化率远小于反应器体积变化率,造成放电产生的等离子体在单位放电反应区域内的分布减少,即能量体积密度变小。综合翘片间距对功率与能量体积密度的影响,A/B=1时的翘片结构为实验最优电极配置。
-
1) 初始质量浓度对VOCs降解的影响。含氯VOCs在不同工业领域的排放浓度不同,因此探究初始质量浓度对降解效果的影响十分重要。图3所示为放电间距为2.4 cm的反应器内,初始气体流量256.8 mL·min−1,含氧量4%,脉冲频率为50 Hz,反应器对二氯甲烷的降解效率及能量效率。随着脉冲峰值电压的增加,注入反应器功率不断增加,二氯甲烷降解效率也呈现上升的趋势。在相同脉冲峰值电压条件下,二氯甲烷降解效率与初始质量浓度成反比,与能量效率成正比。当脉冲峰值电压为31 kV时,初始质量浓度从379.2 mg·m−3增至1 895.8 mg·m−3,二氯甲烷降解效率从42.6%降至33.0%,相应的能量效率从1.23 g·kWh−1升至4.40 g·kWh−1。这是因为保持脉冲峰值电压和输入功率恒定,放电区域产生的高能电子、活性粒子数目基本保持不变。初始浓度越高意味着单位体积内存在于反应器中的污染分子数量越多,而恒定的输入功率产生的高能电子和活性粒子有限,无法满足大量二氯甲烷的降解需求。因此,二氯甲烷的降解效率随之降低[22-23]。同时,较高的初始质量浓度增加了单位体积内高能电子及活性粒子与二氯甲烷碰撞的几率,使得反应器放电区域产生的能量得到更充分利用,从而提高了能量效率[24-25]。
2) 停留时间对VOCs降解的影响。在电极结构确定的反应器内,气体在等离子体放电区域内的停留时间取决于通过反应器的总气体流量。图4为放电间距为2.4 cm的反应器内,二氯甲烷质量浓度为379.2 mg·m−3,含氧量 (体积分数) 为4%,脉冲频率为50 Hz时,气体流量对二氯甲烷降解效率和能量效率的影响。停留时间选取250、214、160.5、128.4和107 s,对应气体体积流量分别为256.8、300、400、500和600 mL·min−1。结果显示,在固定脉冲峰电压条件下,二氯甲烷降解效率随着气体流量的增加呈现下降趋势,但能量效率呈现上升的趋势。在较高的气体流量下,二氯甲烷在等离子体放电区域内停留时间缩短,导致电子碰撞反应和二氯甲烷与自由基、亚稳态等活性粒子的碰撞概率降低,反应不彻底,从而降低了二氯甲烷降解效率[26]。但是随着气体流量的增加,单位时间内通过放电区域的二氯甲烷分子数量增加,在相同的脉冲峰值电压下对能量的利用更加充分,能量效率随之提高。因此,在之后的实验中选择250 s为二氯甲烷的最佳停留时间。
3) 含氧量对VOCs降解的影响。在等离子体处理二氯甲烷过程中,气流含氧量对活性粒子的产生有着重要的影响[13]。氧气小幅度的增加将导致含氧活性粒子增加,从而影响二氯甲烷降解效率[27–29]。图5所示为放电间距为2.4 cm的反应器中二氯甲烷质量浓度为379.2 mg·m−3,气体体积流量256.8 mL·min−1,脉冲频率为50 Hz时,气流含氧量 (体积分数) 对二氯甲烷降解效率和能量效率的影响。随着含氧量的增加,二氯甲烷降解效率呈现下降趋势;当峰值电压为31 kV时,含氧量 (体积分数) 由0增加至10%,二氯甲烷降解效率从56.8%降至9.1%;能量效率也在不断下降,在含氧量 (体积分数) 10%条件下,仅有0.56 g·kWh−1。
ALLAH等[30]发现在填充床反应器中,当含氧量 (体积分数) 为2%~4%,降解效率存在最佳值。HUANG等[31]则发现在脉冲电晕反应器中,二氯甲烷降解效率随着含氧量的增加不断降低,并在纯氮气环境下达到最大值,猜测差异可能是与不同的等离子体放电方式有关。
由于氧气自身具有电负性,会在一定程度上吸附高能电子,使得高能电子与二氯甲烷的直接撞击几率降低,可能导致二氯甲烷降解效率的降低。另外,虽然氧气在电晕放电下易解离产生氧自由基,但氧自由基与二氯甲烷的反应速率远低于氮自由基与二氯甲烷的反应速率[32]。同时,氧气的存在会消耗激部分亚稳态氮原子,从而形成NOx[30],这也会造成降解效率的降低。
4) 脉冲电压峰值对VOCs降解的影响。图6所示为放电间距为2.4 cm的反应器中,二氯甲烷质量浓度为379.2 mg·m−3,气体体积流量256.8 mL·min−1,含氧量 (体积分数) 4%,脉冲频率为50 Hz时,脉冲峰值电压对二氯甲烷降解效率及能量效率的影响。随着脉冲峰值电压的增加,二氯甲烷降解效率不断增加,但能量效率却与之相反,二氯甲烷降解效率在31 kV处取得最大值,为43.3%,对应能量效率为1.23 g·kWh−1。脉冲峰值电压的增加令注入反应器放电功率和放电区域电场强度增加,使高能电子和活性粒子的数量增加,也大大提高了二氯甲烷分子与高能电子及活性粒子间有效碰撞的几率,导致了二氯甲烷降解效率呈现上升趋势[33-35]。同时,随着脉冲峰值电压的增加,能量效率由1.48 g·kWh−1降低至1.23 g·kWh−1。这是因为随着脉冲峰值电压的增加,反应体系中高能电子和活性物质的数量增加,而污染物分子的数量减少,能量以光和热的形式逐渐被消耗,这导致高能电子和活性粒子的利用率降低,所以能量效率随着脉冲峰值电压的增加而不断降低[36-37]。
-
为检测翘片-筒式等离子体反应器降解VOCs的稳定性,进行了二氯甲烷的连续降解实验,结果如图7所示。反应器运行2 h后,二氯甲烷的降解效率呈现下降的趋势,并在之后的连续反应时间内降解效率在35%上下小幅度浮动;此时,反应器注入功率小幅下降,而后依旧保持稳定。这可能是由于反应初始翘片电极表面干净光亮,随着处理时间的增加电极表面有少量气溶胶沉积,影响反应器放电特性,造成注入反应器的功率降低从而使降解效率下降。翘片-筒式反应器在长时间运行后,虽然二氯甲烷降解效率出现了小幅度的波动,但波动前后均呈现稳定状态,且波动后其降解效率稳定在约35%,也显示出其具有较好的稳定性与降解效率。
为观察反应器运行前后翘片电极气溶胶沉积情况,进行了17 h二氯甲烷降解实验。实验条件为:二氯甲烷质量浓度379.2 mg·m−3、气体体积流量256.8 mL·min−1、含氧量 (体积分数) 4%及脉冲频率50 Hz。气溶胶未沉淀前翘片电极结构表面及凹槽处干净光亮(图8(a)) ;而经过17 h放电连续处理后的翘片结构表面及凹槽处明显沉积了一层黄褐色物质(图8(b))。另外,观察到放电产生的气溶胶更容易沉积在翘片结构凹槽处,极少量沉积在翘片表面上。由于翘片边沿为放电区域而凹槽处并非放电区域,气溶胶沉积在凹槽处不会对放电产生影响。
进一步比较气溶胶沉积前后的放电图像发现,气溶胶沉积前 (图8 (c) ) 、后 (图8 (d) ) 放电情况未有明显差异,靠近放电区域都产生明显光晕并伴随明暗交替的细光条。在较低二氯甲烷浓度下,降解产物气溶胶沉积在凹槽处,避免其大量直接沉积在放电区域表面从而影响放电,在一定程度上延长了翘片稳定放电的时间[20]。同时,气溶胶在放电区表面的沉积层较薄,相对较少,对放电影响不大。
-
1) 降解产物分析。使用放电间距为2.4 cm的反应器,二氯甲烷质量浓度为379.2 mg·m−3,气体体积流量500 mL·min−1,在脉冲频率为50 Hz下,对氮气环境和含氧量 (体积分数) 2%条件下二氯甲烷降解产物进行傅里叶红外(FTIR)分析和气相有机产物的气质联用(GC-MS)分析,并使用便携式气体检测仪对排放尾气中的HCl进行了检测,结果如图9所示。在氮气环境中等离子体降解二氯甲烷生成的主要产物是HCl、CO2,检测到含氧物质可能是由于进口中的少量水蒸气及氮气和二氯甲烷气体中的杂质造成的;加入氧气后二氯甲烷降解生成的主要产物是CO2、CO和氮氧化物 (NO2、NO及N2O) ,没有检测到HCl。分析原因可能是其在与∙O、∙OH的反应中被氧化[30]。国内外有不少研究人员也进行过类似的分析和报道。WALLIS等[38]在氮气环境中检测到了HCl、CO2,以及CO、HCN和N2O。ALLAH等[30]在氧气环境中检测到CO2、CO、NO2、NO、N2O,以及COCl2和NOCl。由于本实验采用的FTIR 对于检测样品种类有限,并未检测到HCN、COCl2及NOCl的存在。
图10为等离子体降解二氯甲烷产生的气相有机产物GC-MS谱图。生成的气相有机产物(表1)主要为氯代烃(氯甲烷、三氯甲烷、1,2-二氯乙烷、四氯化碳)、长链烷烃(2-甲基戊烷、正辛烷 、癸烷、3-甲基十一烷、正十五烷)、醇类(乙醇)、醚类(四氢呋喃)和醛类(庚醛、壬醛)。其他研究者也报道过,离子体降解二氯甲烷过程中主要产物为氯代烃。GAIKWAD 等[39]在采用放电等离子体降解二氯甲烷过程中检测到三氯甲烷。SGRO等[40]在二氯甲烷降解产物中发现了氯甲烷。LI等[41]在放电等离子体系统中检测到了氯甲烷、三氯甲烷和四氯化碳。
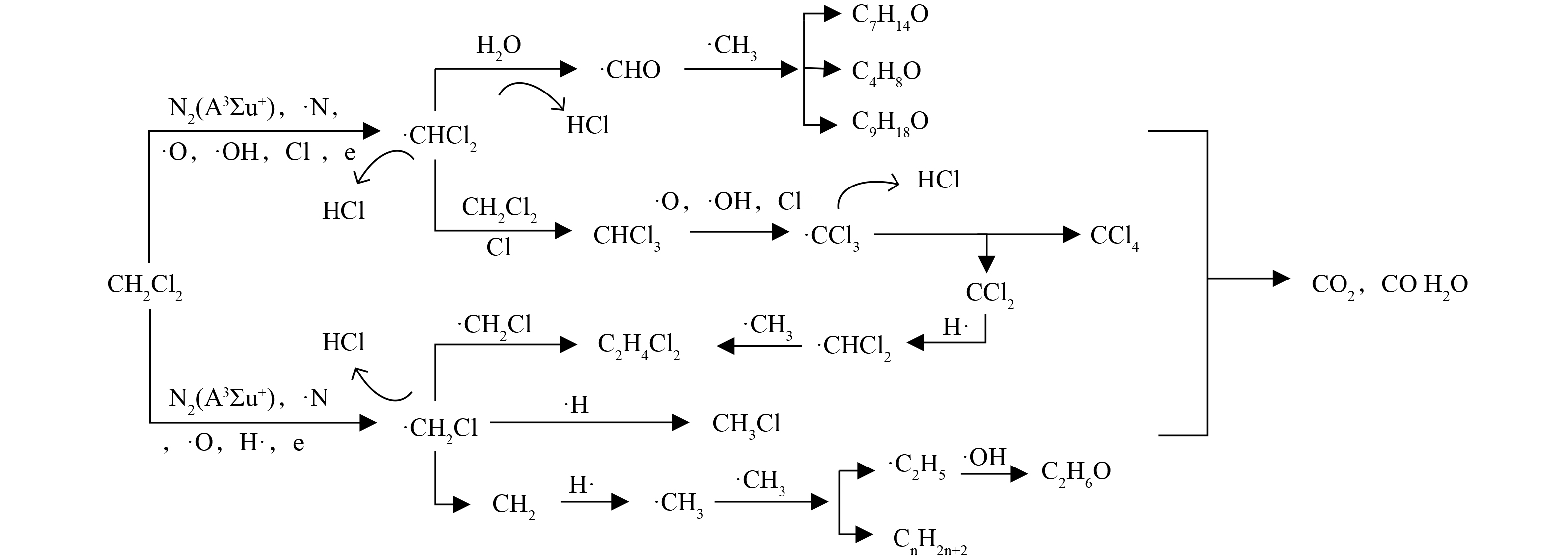
2) 反应路径推测。根据气相有机产物的GC-MS分析,推测翘片-筒式脉冲电晕流光放电降解二氯甲烷的可能路径 (图11) 。等离子体降解二氯甲烷主要依靠活性物质 (N2(A3Σu+)、∙O、∙N、∙OH) 的氧化作用。由于二氯甲烷浓度较低,高能电子与其直接发生碰撞而被降解的几率较小。活性物质可将二氯甲烷氧化分解,此外∙O还能与二氯甲烷反应生成∙OH;另外,高能电子也可直接与二氯甲烷分子发生碰撞,造成C—H及C—Cl键的断裂,生成∙H和Cl−,见式(7)~(13)[42–44]。
在本研究中,二氯甲烷的降解路径主要有2条。第一条路径为二氯甲烷脱氯生成·CH2Cl。·CH2Cl自身相互反应生成1,2-二氯乙烷[11],也可与∙H反应生成氯甲烷[39-41,45],除此之外还能继续脱氯生成亚甲基,亚甲基与∙H反应生成甲基。生成的甲基再与∙OH反应生成乙醇,还可通过自身相互反应生成长链烷烃(2-甲基戊烷、正辛烷 、癸烷、3-甲基十一烷、正十五烷等)。第二条路径是二氯甲烷脱氢生成·CHCl2,CHCl2一方面与H2O作用脱除HCl生成·CHO[11],·CHO再与甲基反应生成四氢呋喃、庚醛和壬醛。另一方面·CHCl2在二氯甲烷或者Cl−的作用下生成三氯甲烷[41,30],部分三氯甲烷也会与活性粒子 (∙O、∙N、Cl−) 反应生成·CCl3,·CCl3继续与氯离子反应生成四氯化碳[41]。另外,高能电子、活性物质将会和二氯甲烷中间体和各种碎片分子等进一步生成各种中间产物,最终活性粒子将中间产物进一步氧化形成CO2。
-
1) 当翘片-筒式脉冲电晕流光反应器相邻翘片间距(A)/放电间距(B)=1,脉冲电压为37 kV的条件下,功率和能量体积密度的最大值分别0.95 W和23.6 mJ·L−1,可同时兼顾功率和能量体积密度,为翘片电极最优配置。
2) 翘片-筒式脉冲电晕流光反应器注入功率在前2 h保持稳定,2 h后功率下降但依旧保持稳定状态,显示出翘片-筒式反应器较好的稳定性与降解效率。放电17 h后在翘片电极结构表面形成气溶胶,大量沉积在翘片结构凹槽处,没有明显影响放电,使得反应器降解性能与系统放电强度能在较长时间内维持稳定。
3) 根据GC-MS产物分析,脉冲电晕流光放电等离子降解二氯甲烷的降解产物主要为氯代烃 (氯甲烷、1,2-二氯乙烷、四氯化碳、三氯甲烷) 、长链烷烃以及醛类等。二氯甲烷的降解依靠高能电子碰撞与活性物质的氧化作用进行。脱氢、脱氯是导致二氯甲烷C-H及C-Cl键的断裂的主要过程,而活性粒子的氧化作用是导致二氯甲烷进一步降解的主要途径。
翘片-筒式脉冲电晕流光放电装置对二氯甲烷的降解效果及其稳定性
Degradation effect and system stability of dichloromethane by fins-cylinder pulse corona streamer discharge
-
摘要: 传统结构线-筒式电晕放电装置在降解VOCs时生成的副产物会粘附在放电区域(线电极)上,使得放电间距减小,导致降解效果不稳定。翘片-筒式脉冲电晕流光放电等离子装置的电极结构可将放电区域(翘片尖端-筒)与副产物粘附区域(连接翘片的轴线)分开,从而维持装置的放电强度和稳定性。当翘片-筒式脉冲电晕流光放电反应器相邻翘片间距/放电间距为1,在37 kV下,反应器功率为0.95 W,能量体积密度为和23.6 mJ·L−1,为最优电极配置;处理二氯甲烷2 h后的降解效率呈现小幅度下降后稳定在35%。因此,放电形成的气溶胶部分沉积在凹槽处,可维持反应器稳定的放电强度及对二氯甲烷的降解效率。本研究结果可为低温等离子体处理VOCs废气的性能提升提供参考。Abstract: The conventional structure of corona discharge is mainly wire-cylinder, and the by-products generated during the degradation of VOCs will adhere to the discharge area (wire electrode) to reduce the discharge spacing, resulting in unstable degradation effect. Therefore, this study proposed a fins-cylinder pulsed corona streamer discharge plasma electrode structure, which separated the discharge area (tip of fins-cylinder) from the by-product adhesion area (axis connecting the fins)to enhance the system discharge intensity and stability. The results showed that the fins electrode structure with adjacent fin spacing/discharge spacing (A/B=1) was the best electrode design, with highest values of reactor power and energy volume density at 31 kV being 0.98 W and 37.9 mJ·L−1, respectively. The degradation efficiency of dichloromethane decreased slightly after 2 h treatment and then stabilized at 35%. Aerosols produced by the discharge were partially deposited in the grooves without substantially altering the electrode discharge, which guaranteed the stability of the intensity of the discharge and the degradation efficiency of dichloromethane.. This study can serve as a foundation for future research on low temperature plasma treatment of VOC exhaust gas.
-
Key words:
- fins-cylinder /
- corona streamer discharge /
- plasma /
- degradation /
- volatile organic compounds(VOCs)
-

-
表 1 气相有机产物组成信息表
Table 1. Information on the composition of gaseous organic products
名称 时间/min 分子式 匹配度 氯甲烷 5.34 CH3Cl 98.5% 乙醇 6.63 C2H6O 99.6% 二氯甲烷 8.20 CH2Cl2 99.1% 2-甲基戊烷 9.98 C6H14 95.4% 三氯甲烷 11.28 CHCl3 93.9% 四氢呋喃 11.82 C4H8O 97.4% 1,2-二氯乙烷 12.27 C2H4Cl2 91.0% 四氯化碳 13.54 CCl4 99.3% 正辛烷 19.64 C8H18 97.4% 庚醛 22.84 C7H14O 95.0% 癸烷 27.32 C10H22 96.3% 壬醛 30.49 C9H18O 90.1% 3-甲基十一烷 34.49 C12H26 97.3% 3-甲基十一烷 34.49 C12H26 97.3% -
[1] VANDENBROUCKE A M, MORENT R, Geyter D N, et al. Non-thermal plasmas for non-catalytic and catalytic VOC abatement[J]. Journal of Hazardous Materials, 2011, 195: 30-54. doi: 10.1016/j.jhazmat.2011.08.060 [2] SCHIAVON M, TORRETTA V, CASAZZA A, et al. Non-thermal Plasma as an Innovative Option for the Abatement of Volatile Organic Compounds: a Review[J]. Water, Air, & Soil Pollution, 2017, 228(10): 388. [3] Durme V J, DEWULF J, LEYS C, et al. Combining non-thermal plasma with heterogeneous catalysis in waste gas treatment: A review[J]. Applied Catalysis B:Environmental, 2008, 78(3-4): 324-333. doi: 10.1016/j.apcatb.2007.09.035 [4] LI Q Q, SU G J, LI C Q, et al. Emission profiles, ozone formation potential and health-risk assessment of volatile organic compounds in rubber footwear industries in China[J]. Journal of Hazardous Materials, 2019, 375: 52-60. doi: 10.1016/j.jhazmat.2019.04.064 [5] ONDARTS M, HAJJI W, OUTIN J, et al. Non-Thermal Plasma for indoor air treatment: Toluene degradation in a corona discharge at ppbv levels[J]. Chemical Engineering Research and Design, 2017, 118: 194-205. doi: 10.1016/j.cherd.2016.12.015 [6] NGUYEN V T, NGUYEN D B, MOK Y S, et al. Removal of ethyl acetate in air by using different types of corona discharges generated in a honeycomb monolith structure coated with Pd/γ-alumina[J]. Journal of Hazardous Materials, 2021, 416: 126162. doi: 10.1016/j.jhazmat.2021.126162 [7] 李娜, 彭邦发, 姜楠, 等. 气液混合脉冲放电再生吸附苯酚的活性炭[J]. 化工环保, 2022, 42(5): 592-597. doi: 10.3969/j.issn.1006-1878.2022.05.013 [8] 商莹, 姜楠, 孙浩洋, 等. 脉冲放电等离子体同时处理地下水中Cr(VI)和阿特拉津的研究[J]. 环境科学学报, 2022, 42(7): 371-379. doi: 10.13671/j.hjkxxb.2021.0560 [9] MA F J, ZHU Y N, WU B, et al. Degradation of DDTs in thermal desorption off-gas by pulsed corona discharge plasma[J]. Chemosphere, 2019, 233: 913-919. doi: 10.1016/j.chemosphere.2019.05.292 [10] ZHANG X M, FENG F D, LI S R, et al. Aerosol formation from styrene removal with an AC/DC streamer corona plasma system in air[J]. Chemical Engineering Journal, 2013, 232: 527-533. doi: 10.1016/j.cej.2013.08.009 [11] JOSE J, RAMANUJAM S, PHILIP L. Applicability of pulsed corona discharge treatment for the degradation of chloroform[J]. Chemical Engineering Journal, 2019, 360: 1341-1354. doi: 10.1016/j.cej.2018.10.199 [12] KIM H H. Nonthermal plasma processing for air-pollution control: A historical review, current issues, and future prospects[J]. Plasma Processes and Polymers, 2004, 1(2): 91-110. doi: 10.1002/ppap.200400028 [13] XIAO G, XU W P, WU R B, et al. Non-thermal plasmas for VOCs abatement[J]. Plasma Chemistry and Plasma Processing, 2014, 34(5): 1033-1065. doi: 10.1007/s11090-014-9562-0 [14] YAO X M, JIANG N, LI J, et al. An improved corona discharge ignited by oxide cathodes with high secondary electron emission for toluene degradation[J]. Chemical Engineering Journal, 2019, 362: 339-348. doi: 10.1016/j.cej.2018.12.151 [15] ZHANG H B, LI K, SUN T H, et al. The removal of styrene using a dielectric barrier discharge (DBD) reactor and the analysis of the by-products and intermediates[J]. Research on Chemical Intermediates, 2013, 39(3): 1021-1035. doi: 10.1007/s11164-012-0664-0 [16] KARATUM O, DESHUSSES M A. A comparative study of dilute VOCs treatment in a non-thermal plasma reactor[J]. Chemical Engineering Journal, 2016, 294: 308-315. doi: 10.1016/j.cej.2016.03.002 [17] 李杰, 刘林茂. 采用V型极板收集高比电阻粉尘的实验研究[J]. 环境科学, 1995, 16(6): 33-35. doi: 10.3321/j.issn:0250-3301.1995.06.002 [18] JIANG N, ZHAO Y H, SHANG K F, et al. Degradation of toluene by pulse-modulated multistage DBD plasma: key parameters optimization through response surface methodology (RSM) and degradation pathway analysis[J]. Journal of Hazardous Materials, 2020(393): 122365. [19] JIANG N, LI J, HU L, et al. Optimization investigation on geometrical parameters of a multistage asymmetric fin-type DBD reactor for improved degradation of toluene [J]. International Journal of Plasma Environmental Science and Technology. 2020(14): e03002 [20] 翟旺生. 非对称杆式介质阻挡放电及其耦合催化条件下对甲苯的降解研究[D]. 大连: 大连理工大学, 2019. [21] 胡露. 翅片杆-杆介质阻挡放电协同催化降解甲硫醚的研究[D]. 大连: 大连理工大学, 2021. [22] LIU R Q, SONG H, LI B, et al. Simultaneous removal of toluene and styrene by non-thermal plasma-catalysis: Effect of VOCs interaction and system configuration[J]. Chemosphere, 2021, 263: 127893. doi: 10.1016/j.chemosphere.2020.127893 [23] LI S J, DANG X Q, YU X, et al. The application of dielectric barrier discharge non-thermal plasma in VOCs abatement: A review[J]. Chemical Engineering Journal, 2020, 388: 124275. doi: 10.1016/j.cej.2020.124275 [24] ZHU R Y, MAO Y B, JIANG L Y, et al. Performance of chlorobenzene removal in a nonthermal plasma catalysis reactor and evaluation of its byproducts[J]. Chemical Engineering Journal, 2015, 279: 463-471. doi: 10.1016/j.cej.2015.05.043 [25] ZHU X B, TU X, MEI D H, et al. Investigation of hybrid plasma-catalytic removal of acetone over CuO/γ-Al2 O3 catalysts using response surface method[J]. Chemosphere, 2016, 155: 9-17. doi: 10.1016/j.chemosphere.2016.03.114 [26] DU C M, HUANG Y N, GONG X J, et al. Plasma purification of halogen volatile organic compounds[J]. IEEE Transactions on Plasma Science, 2018, 46(4): 838-858. doi: 10.1109/TPS.2018.2808243 [27] 李国平. 脉冲电晕结合催化进行甲烷氯化和二氯甲烷降解实验研究[D]. 杭州: 浙江工业大学, 2014. [28] 任翔宇. 双介质阻挡放电耦合气升式生物填料反应器处理甲苯和二氯甲烷的工艺研究[D]. 杭州: 浙江大学, 2018. [29] 陈安格. 旋转电极放电反应器去除三氯乙烯和二氯甲烷实验研究[D]. 杭州: 浙江工业大学, 2020. [30] ALLAH Z A, WHITEHEAD J C, MARTIN P. Remediation of dichloromethane (CH2Cl2) using non-thermal, atmospheric pressure plasma generated in a packed-bed reactor[J]. Environmental Science & Technology, 2014, 48(1): 558-565. [31] HUANG L, NAKAJO K, OZAWA S, et al. Decomposition of dichloromethane in a wire-in-tube pulsed corona reactor[J]. Environmental Science & Technology, 2001, 35(6): 1276-1281. [32] PENETRANTE B M, HSIAO M C, BARDSLEY J N, et al. Decomposition of methylene chloride by electron beam and pulsed corona processing[J]. Physics Letters A, 1997, 235(1): 76-82. doi: 10.1016/S0375-9601(97)00611-7 [33] 郑毅文. 介质阻挡放电协同催化降解邻二氯苯的实验研究[D]. 杭州: 浙江大学, 2021. [34] NGUYEN V T, YOON K H, MOK Y S, et al. Practical-scale honeycomb catalytic reactor coupled with non-thermal plasma for high-throughput removal of isopropanol[J]. Chemical Engineering Journal, 2022, 430: 132905. doi: 10.1016/j.cej.2021.132905 [35] MA T P, CHEN J Y, LIU J Q, et al. Promotion of toluene degradation in negative DC corona discharge by magnetic field[J]. Journal of Physics D:Applied Physics, 2018, 51(42): 425203. doi: 10.1088/1361-6463/aadcd2 [36] LI S J, LI Y, YU X, et al. A novel double dielectric barrier discharge reactor for toluene abatement: Role of different discharge zones and reactive species[J]. Journal of Cleaner Production, 2022, 368: 133073. doi: 10.1016/j.jclepro.2022.133073 [37] 杨仕玲. 低温等离子体-纳米后催化协同降解挥发性有机化合物基础研究[D]. 杭州: 浙江大学, 2020. [38] WALLIS A E, WHITEHEAD J C, ZHANG K. The removal of dichloromethane from atmospheric pressure nitrogen gas streams using plasma-assisted catalysis[J]. Applied Catalysis B:Environmental, 2007, 74(1-2): 111-116. doi: 10.1016/j.apcatb.2006.11.020 [39] GAIKWAD V, KENNEDY E, MACKIE J, et al. Reaction of dichloromethane under non-oxidative conditions in a dielectric barrier discharge reactor and characterisation of the resultant polymer[J]. Chemical Engineering Journal, 2016, 290: 499-506. doi: 10.1016/j.cej.2015.12.105 [40] SGRO L A, KOSHLAND C P, LUCAS D, et al. Postflame reaction chemistry of dichloromethane: variations in equivalence ratio and temperature[J]. Combustion and Flame, 2000, 120(4): 492-503. doi: 10.1016/S0010-2180(99)00119-4 [41] LI C T, LEE W J, CHEN C Y. CH2Cl2 Decomposition by Using a Radio-Frequency Plasma System[J]. Journal of Chemical Technology and Biotechnology, 1996, 66: 382-388. doi: 10.1002/(SICI)1097-4660(199608)66:4<382::AID-JCTB518>3.0.CO;2-C [42] WAllIS A E, WHITEHEAD J C, ZHANG K. The removal of dichloromethane from atmospheric pressure air streams using plasma-assisted catalysis[J]. Applied Catalysis B:Environmental, 2007, 72(3/4): 282-288. [43] FAN X, KANG S J, LI J, et al. Formation of nitrogen oxides (N2O, NO, and NO2) in typical plasma and plasma-catalytic processes for air pollution control[J]. Water, Air, & Soil Pollution, 2018, 229(11): 351. [44] FITZSIMMONS C, ISMAIL F, WHITEHEAD J C, et al. The chemistry of dichloromethane destruction in atmospheric-pressure gas streams by a dielectric packed-bed plasma reactor[J]. The Journal of Physical Chemistry A, 2000, 104(25): 6032-6038. doi: 10.1021/jp000354c [45] HO W P, BARAT R B, BOZZELLI J W. Thermal reactions of CH2Cl2 in H2/O2 mixtures: implications for chlorine inhibition of CO conversion to CO2[J]. Combustion and Flame, 1992, 88: 265-295. doi: 10.1016/0010-2180(92)90035-N -




 下载:
下载: