-
室外空气污染制定的标准和法规,已显著降低了全球许多地区的颗粒物、氮氧化物和二氧化硫的排放量。然而,室内空气污染却未能受到同样关注,且室内空气污染的相关科学研究进展也少于室外空气污染,使政府难以制定有针对性的政策和控制措施,室内空气质量成为影响人类健康的重要因素[1]。挥发性有机物是危害人类健康的“杀手”,而甲醛是一种典型的挥发性有机气体,室内装修甚至烹饪过程都成为甲醛释放的重要来源[2-3]。短时间接触甲醛会产生慢性中毒、皮肤刺激等症状,而长期接触或生活在甲醛超标环境中会严重刺激呼吸系统,导致慢性呼吸道疾病,甚至癌变[4]。因此尤其针对室内低浓度甲醛治理,开发一种安全有效的甲醛净化方法对人类健康至关重要。
目前,物理吸附[5]、光催化降解[6-7]、热催化降解[8-9]等甲醛分解技术已在相关研究领域取得显著效果,其中光催化降解在室温下将甲醛氧化成二氧化碳和水,具有高效、稳定、无二次污染的特点,被认为是一种高级氧化工艺[10]。常见的催化剂主要是氧化物和硫化物半导体。其中,ZnO因无毒、环保、廉价易得[11],同时具有激子结合能高 (60 meV) 等优点而被广泛应用于光电转化、太阳能电池、光催化等领域[12-15]。然而,单一ZnO带隙宽、电子空穴分离程度低等不足制约其在光催化领域的实际应用[16-18],一般而言,ZnO与窄带隙半导体构建异质结有利于解决其光吸收范围窄的弊端[19-20]。CdS作为一种典型的窄带隙半导体因具有强烈的可见光活性及制备成本较低等特点受到广泛关注[21]。半导体ZnO和CdS复合可提高电子-空穴对的分离效率并拓宽光吸收范围,进而提升ZnO的光催化活性[22]。例如LU等[23]通过水热合成簇状CdS/ZnO纳米管S型异质结用于光催化产氢,促进了电子-空穴对的分离,显著提高了单一组分的光催化效率。
然而,半导体表面没有高活性的还原位点,限制了催化剂的催化活性,通常引入助催化剂来解决,助催化剂的功能多样性可有效促进催化活性的提升[24]。贵金属助催化剂因自身良好的光学活性备受瞩目[25-27],其具有的可见光活性可充分利用太阳能,产生表面等离子共振效应 (SPR) ,诱导捕获电子用于光催化反应,具体表现为当光入射到贵金属表面,贵金属内部电子发生均匀、快速、高效的等离子体加热,产生大量热电子,并转移至半导体表面[28],从而提高光学性能。例如,Au纳米颗粒修饰的TiO2纳米棒具有较高的光催化活性,Au纳米颗粒的引入促进热电子向TiO2表面迁移,提高半导体对可见光的利用和电子的捕获能力[29]。YU等[30]研究Au纳米颗粒的表面等离子共振效应提高半导体异质结的光催化活性,结果表明当Au纳米颗粒受到光照射后,表面电子会转移到异质结中用于参与反应,使得用于光催化反应的电子数量大大增加。
因此,本研究首先使用两步水热法在衬底表面制备了一层ZnO/CdS粉末薄膜,然后利用化学浴沉积法,在ZnO/CdS异质结表面修饰一层金纳米颗粒,用于低质量浓度甲醛去除研究,并通过循环稳定性实验验证了催化剂在应用方面的潜力。此外,还探究了催化剂种类、光照波长、甲醛初始质量浓度、相对湿度对甲醛去除效果的影响,为室温下低质量浓度甲醛的实际应用提供参考。
-
无水乙醇 (CH3CH2OH ) 、氨水 (NH3·H2O) 、硝酸锌 (Zn(NO3)2) 、丙酮 (CH3COCH3) 、四水硝酸镉 (Cd(NO3)2·4H2O) 、硫代乙酰胺 (C2H5NS) 、四水氯金酸 (HAuCl4·4H2O) 、二水柠檬酸钠 (Na3C6H5O7·2H2O) 均为分析纯;甲醛溶液 (HCHO,体积分数38%) 。
X射线衍射仪(X'TRA型,美国赛默飞世尔科技有限公司);扫描电子显微镜 (Nova NanoSEM450,美国FEI 公司) ;高分辨率透射电镜 (X-MaxN 80T IE250,日本捷欧路公司) ; X射线光电子能谱仪(PHI5000 VersaProbe型,日本ULVAC-PHI公司);电感耦合等离子体质谱 (ICP-MS,7500CE,美国安捷伦科技有限公司) ;荧光光谱仪 (Shimadzu RF-5301,日本岛津公司) ;电化学工作站 (CHI-660E,武汉科思特仪器股份有限公司) ;甲醛分析仪(4160-19.99m型,美国INTERSCAN公司)。
-
取0.654 4 g Zn(NO3)2·2H2O 分散到40 mL去离子水中,超声形成均相后,缓慢滴加氨水至溶液的pH=10~11并不断搅拌20 min。为了固定催化剂粉末和测试催化剂循环使用的稳定性,实验将采用载玻片作为衬底,使催化剂在其表面生长。将载玻片 (2 cm×2 cm) 以45°斜放入上述溶液中,将装有混合溶液和载玻片的玻璃瓶转移至烘箱中,在95 ℃下反应3 h,自然降至室温后静置48 h,取出衬底后使用去离子水和无水乙醇各清洗3次,并在80 ℃下干燥2 h,最后将样品置于马弗炉中,以2 ℃·min−1升温速度从室温升至350 ℃,并保温2 h,得到ZnO。
取0.381 4 g Cd(NO3)2·4H2O和0.148 5 g C2H5NS置于70 mL去离子水中混合并连续搅拌形成均相,然后将已制备的ZnO (0.040 7 g) 分散在混合溶液中并转移至高压反应釜中,将反应釜移至140 ℃烘箱中反应4 h,降至室温后,收集沉淀并用去离子水和无水乙醇清洗3次,然后置于80 ℃烘箱中干燥2 h,最后将样品置于马弗炉中,以2 ℃·min−1升温速度从室温升至350 ℃,并保温2 h,得到ZnO/CdS 纳米棒,记为ZC。
使用化学浴沉积法将Au纳米颗粒修饰在ZnO和ZnO/CdS纳米棒外侧,具体步骤:取1 mL HAuCl4·4H2O溶液(0.05 mol·L−1)加入100 mL去离子水中,并不断均匀搅拌20 min,然后加入 0.160 2 g Na3C6H5O7·2H2O并搅拌30 min,将ZnO和ZnO/CdS催化剂 (各1g) 分别分散在上述混合溶液中,并在95 ℃的黑暗条件下放置35 min,得到样品ZnO/Au和ZnO/CdS/Au,标记为ZAu和ZCAu。
-
在1 m3密闭反应舱内对各种催化剂进行甲醛去除测试。将尺寸为2 cm×2 cm的负载有催化剂的载玻片 (3片) 放入到反应舱内,关闭舱门,然后使用干燥器和注射去离子水的方法调节反应舱内湿度 (20%~80%) 。在湿度稳定后,使用微量进样针向反应舱内注射一定量的甲醛溶液(38%),开启风扇使甲醛完全挥发,反应舱内甲醛质量浓度约为1.25~5 mg·m−3;将甲醛测试仪进气、出气口分别连接到舱上的气体通道,间歇式分析舱内甲醛质量浓度。
-
通过场发射扫描电子显微镜 (FESEM) 观察样品的形貌结构。水热法制备的ZnO呈现为直径约500 nm,长度约2 μm的纳米棒组成的花簇型三维结构 (图1(a)) 。修饰的CdS和Au纳米颗粒,均匀覆盖在ZnO纳米棒表面,形成复合材料ZCAu (图1(b)) 。
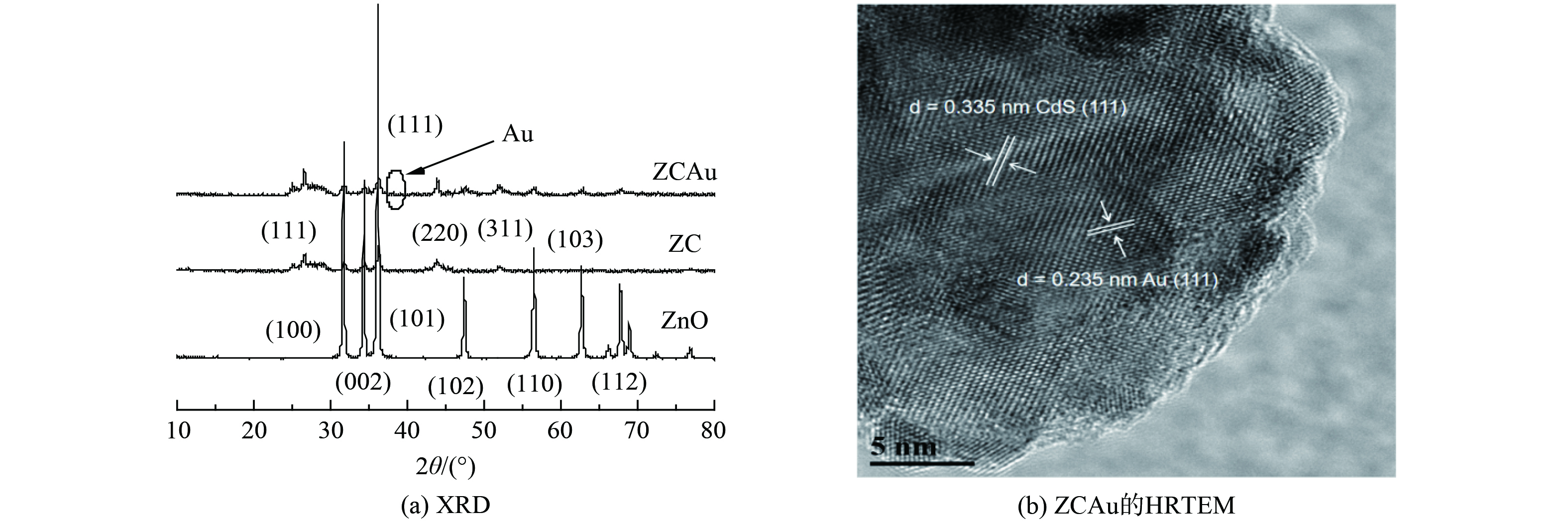
ZnO、ZC和ZCAu 的X射线衍射 (XRD) 图谱如图2(a)所示,ZnO存在6个明显的特征峰,分别对应六方纤锌矿ZnO (JCPDS NO.36-1451) 的 (100) , (002) , (101) , (102) , (110) 和 (112) 面。此外, (101) 面具有最强的衍射峰,说明ZnO 纳米棒主要沿该方向生长,负载CdS后,增加3个显著的衍射峰,分别属于立方相CdS (JCPDS No. 65-2887) 的 (111) 、 (220) 和 (311) 面,说明CdS成功负载在ZnO 纳米棒表面[23]。ZCAu纳米棒的XRD图谱显示在38.28°处有1个弱衍射峰,对应立方相Au的 (111) 面 (JCPDS NO.02-1095) ,此结果说明Au纳米颗粒成功修饰在ZC 纳米棒表面[31]。由于助催化剂相对含量较少,Au的衍射峰强度较弱。
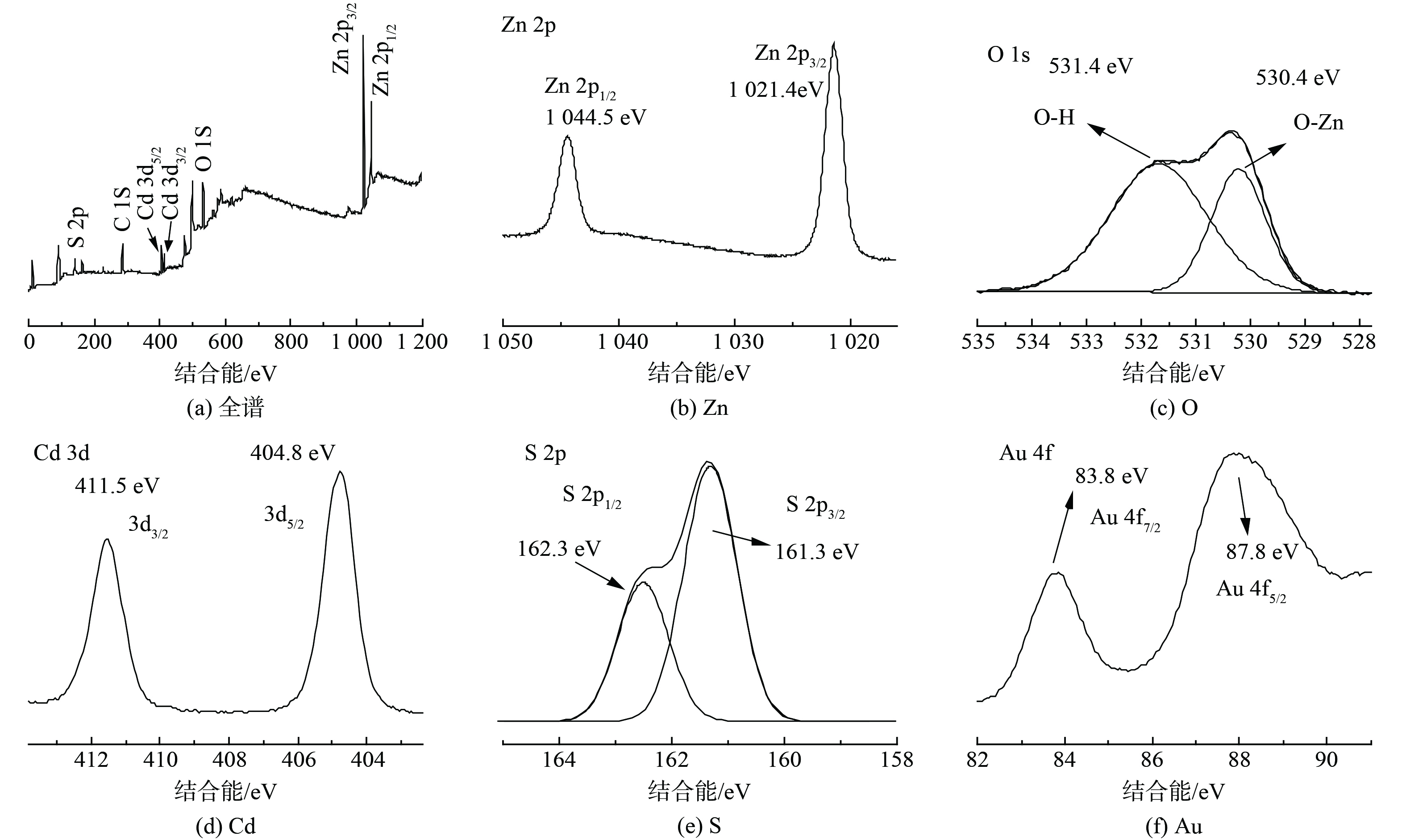
为进一步表征Au的存在,采用高分辨率透射电镜 (HRTEM) 表征样品的微观形貌和晶格结构,图2(b)中显示的区域为纳米棒的外侧,可观察到CdS和Au纳米颗粒,CdS的 (111) 面晶格间距为0.335 nm;Au的 (111) 面晶格间距为0.235 nm,CdS和Au的分界面不明显说明Au纳米颗粒紧密生长在CdS上。ZCAu异质结构的元素化学价态通过X射线光电子能谱 (XPS) 进行表征。主要元素的结合能数据如图3(a)所示,样品存在Zn、O、Cd、S和Au元素。图3(b)为Zn 2p图谱,显示其由2p1/2和2p3/2两种轨道组成,峰位置分别位于1021.4 eV和1044.5 eV,计算出自旋轨道分裂峰的能量差为23.1 eV,说明了Zn在化合物中为+2价态,与ZnO中的Zn2+吻合。图3(c)显示了不对称的O 1s峰,其来源可分为2种,一是ZnO中Zn原子的晶格氧 (Zn-O) (530.0 eV) ,另一个是空穴表面羟基中的氧 (531.2 eV) 。Cd 3d图谱如图3(d)所示,存在Cd 3d5/2和3d3/2两种轨道,对应峰位置是404.9 eV和411.6 eV,自旋轨道分裂峰的能量差为6.7 eV,与CdS中的Cd元素一致,说明Cd以+2价态存在。图3(e)显示了S 2p峰,其由S 2p1/2和S 2p3/2轨道组成,对应结合能位置在161.7 eV和162.7 eV,结果说明S以-2价态存在[32]。Au的峰位置分别在83.8 eV和87.8 eV处,对应于Au 4f7/2和Au 4f5/2,表明Au纳米颗粒被成功还原 (图3(f)) [33]。综上表明,XPS进一步证明所制备样品中含有元素的价态和峰位置与ZnO、CdS和Au一致。
为进一步了解光催化剂实际元素占比量,通过电感耦合等离子体质谱 (ICP-MS) 来检测异质结ZCAu的元素绝对含量。如表1所示,Au质量分数为0.8%,Cd元素的质量分数为47.3%,Zn元素质量分数为30.8%,并且与理论配比 (摩尔比ZnO∶CdS=1∶1) 接近,误差由实验过程中样品的转移和制备消耗所造成。
样品的光捕获能力如图4(a)紫外可见吸收光谱 (UV-vis) 所示。ZnO在385 nm处存在强烈的光吸收阈值,这与其本征吸收有关。ZC 纳米棒表现出增强的可见光吸收 (420~525 nm) ,这是由于CdS具有优异的可见光捕获能力,拓宽了复合结构的光吸收范围,结合XRD和HRTEM数据也说明两种半导体形成了异质结。随后将Au纳米颗粒修饰在ZC纳米棒表面,所制备的ZCAu 纳米棒复合结构的光吸收范围进一步红移,这与Au纳米颗粒在可见光下具有卓越的光响应能力有关,同时,由于Au纳米颗粒的等离子体共振效应诱导Au纳米颗粒和CdS电子相互作用,在500~800 nm处有显著的可见光吸收活性[34]。一般来说,样品的荧光光谱 (PL) 信号越强,代表光生电子-空穴对的复合速率越快[35],如图4(b)所示,纯ZnO的PL信号最强,表明其电子-空穴对的复合速率最快,CdS修饰以后,其荧光强度明显减小,说明异质结的构建提高了ZnO的光催化能力,Au纳米颗粒修饰ZC异质结后,PL信号强度进一步减小,表明Au的引入进一步抑制了电子-空穴对的复合,Au本身的等离子体共振效应使光照射后产生的热电子发生转移,用于光催化还原反应的电子数量增多,提高了光催化能力。此外,界面处形成高效的电子转移方式也是光学性能显著提高的原因。
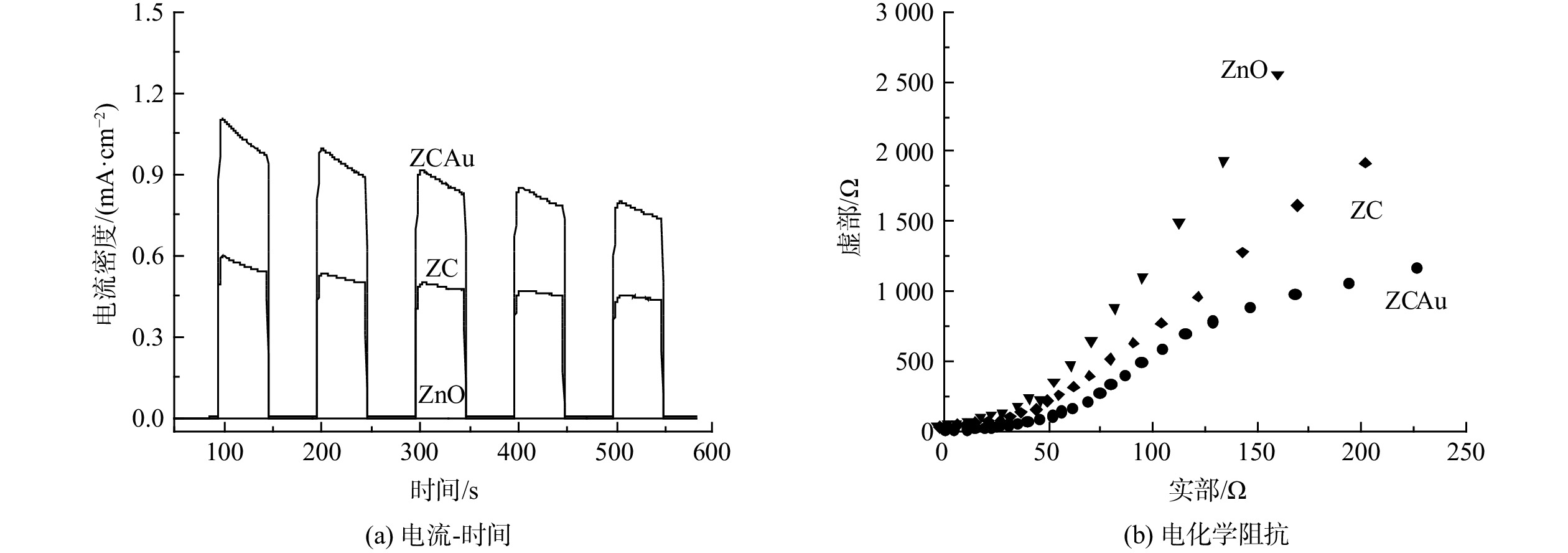
使用电化学工作站进行光电性能测试,研究材料光电响应活性。将已制备好的ZnO,ZC和AuZC光催化剂置于含0.25 mol·L−1 Na2S和0.35 mol·L−1 Na2SO3混合溶液的三电极系统中。光电活性图谱结果如图5所示,图5(a)中光电流强度为光照射瞬时光电流响应,光源关闭时光电流瞬间消失,说明材料中光生电子对光响应灵敏。ZnO的光电流密度小是由于其主要吸收紫外光。ZC异质结显示出较高的光电流密度 (0.5 mA·cm−2) ,表明异质结界面的相互作用提高了材料的光电性能,而Au纳米颗粒修饰ZC后,其光电流密度约为0.9 mA·cm−2,大于ZC光电流强度,远大于ZnO,说明引入Au纳米颗粒进一步提高了催化剂光电性能。载流子的运输和分离效率使用电化学阻抗谱 (EIS) 评估。通常来说,EIS谱线的半圆弧大小代表光电转化阻抗大小,半径越小说明电荷转移的电阻越小,具有更快的载流子运输速率和更高的电子-空穴对分离效率[36]。该EIS实验设定频率为10 KHz到0.1 Hz,图5(b)为奈奎斯曲线 (NIS) ,ZCAu具有最小的圆弧半径, Au纳米颗粒修饰的复合结构具有最小载流子转移阻抗。以上结果表明Au的修饰促进了电荷的分离和运输。
-
1) 不同催化剂种类对甲醛去除的影响。图6(a)显示不同催化剂对甲醛去除的影响。反应舱内甲醛质量浓度控制在1.25 mg·m−3,反应温度为室温,相对湿度保持在40%,光源波长为254 nm,经过2 h反应,对应催化剂ZnO、ZAu、ZC和ZCAu,舱内甲醛质量浓度从1.25 mg·m−3分别下降到0.988、0.86、0.253、0.018 mg·m−3,其中ZCAu对甲醛降解效率最快,ZC次之。以下探究影响因素的对比实验均采用ZCAu为催化剂。
2) 不同光源对甲醛去除的影响。图6(b)为使用不同波长光源 (254、365、460 nm和模拟太阳光) 时,ZCAu对甲醛的去除效果。反应舱内甲醛质量浓度控制在1.25 mg·m−3,反应温度为室温,相对湿度保持在40%。从图中看出,光源波长分别为365和254 nm时,经过2 h反应后甲醛质量浓度从1.25 mg·m−3分别下降到0.153、0.018 mg·m−3。254 nm波长的紫外光对甲醛的降解效率最快,推测这是由于ZnO的光吸收小于400 nm,更容易被254 nm的光激发。且通过实验发现,ZCAu在可见光 (460 nm) 下对甲醛仍具有较高的去除效果,使甲醛质量浓度从1.25 mg·m−3下降到0.528 mg·m−3。此外,光催化降解的一个重要研究方向是实现多波长的太阳光利用,尤其是可见光部分,为此利用氙灯 (300~800 nm) 作为模拟太阳光源进行甲醛去除实验,反应2 h后甲醛质量浓度从1.25 mg·m−3下降至0.015 mg·m−3,表明ZCAu在可见光下对甲醛具有优异的去除效果。为明确催化剂在去除甲醛中的作用,设置了不同光源照射下无催化剂的空白实验,甲醛质量浓度基本无变化,结果进一步说明是催化剂本身起到催化降解的作用。
3) 相对湿度对甲醛去除的影响。空气中水含量对光催化降解甲醛的效果有重要影响。吸附在催化剂上的水分子可以被空穴氧化形成活性羟基,参与到甲醛氧化过程,并不断补充反应过程中消耗的表面羟基,保证高效催化活性。但是水分子会和甲醛分子竞争催化活性中心,过量的水分会引起甲醛去除活性下降[37]。相对湿度变化对ZCAu的甲醛去除影响如图7(a)所示。反应舱内的相对湿度分别控制为20%、40%、60%和80%,反应温度为室温,光源波长为254 nm,经过2 h反应甲醛质量浓度下降至0.705、0.018、0.428、0.818 mg·m−3,表明相对湿度变化对复合材料的甲醛去除活性影响较大,且最适宜的相对湿度为40%。
4) 甲醛起始质量浓度对去除效果的影响。甲醛起始质量浓度对甲醛去效果的影响如图7(b)所示。以ZCAu为光催化剂,反应温度为室温,相对湿度保持在40%,光源波长为254 nm,反应舱内甲醛质量浓度分别控制在1.25、2.5、3.75、5 mg·m−3。由图可知,反应2 h后舱内甲醛的质量浓度分别为0.018、0.023、0.433、1.825 mg·m−3,对应的降解率分别为98.6%,99.1%,88.47%和63.5%。起始质量浓度对复合材料的甲醛去除有显著影响,催化剂对甲醛去除效果随着质量浓度的增大呈现出先增大后减小的趋势。起始质量浓度为2.5 mg·m−3时,甲醛去除效率较快,但随着反应质量浓度进一步提高,甲醛去除效果增速放缓,推测可能原因是反应舱内甲醛质量浓度较低,仍存在部分反应活性位点未参与反应,当甲醛质量浓度高于一定值后,反应活性位点不足,且水分子会和甲醛分子竞争催化活性中心导致催化效率变缓。
5) 稳定性测试。ZCAu每次使用后,在254 nm光照4 h,使表面吸附的甲醛完全氧化分解,以恢复其去除甲醛的活性。表2显示了ZCAu重复次数和甲醛去除效果的关系。经过8次使用,该复合材料的甲醛去除性能没有发生明显变化,并且催化剂质量只脱落了0.052 g,表明其具有优异的多次重复使用性和稳定性。
-
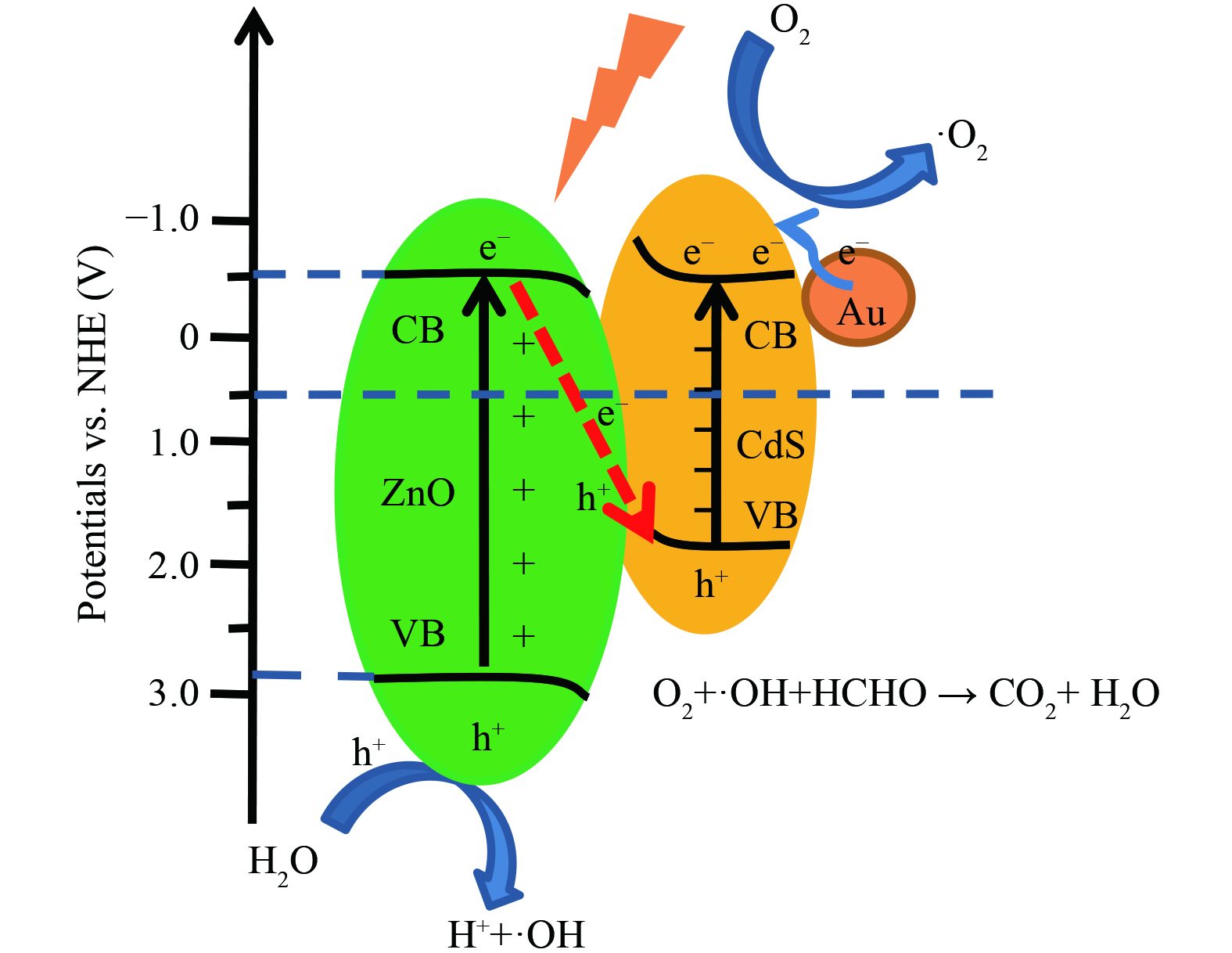
如图8所示,在光照条件下,ZnO和CdS (111) 面接触,各自的电子被激发后从价带跃迁到导带上,随后在价带处留下空穴[23],在电子-空穴对复合过程中,由于内建电场的作用,ZnO 导带上的电子转移到CdS的价带中,CdS导带上发生还原反应, ZnO价带处发生氧化反应,同时Au表现出的等离子共振效应促进热电子注入到相邻的CdS导带中,使导带处积累大量电子。Au的引入进一步提高了异质结光诱导捕获电子能力,从而提高了光催化效率[28,34]。ZnO价带上的空穴和CdS导带上的电子与其粒子表面的溶解氧和水进行反应,生成了具有强氧化能力的超氧自由基 (·O2) 和羟基自由基 (·OH) ,可以将空气中甲醛分子进行催化,使甲醛最终降解为二氧化碳和水分子[2]。
-
1) ZnO/CdS/Au具有较高的甲醛去除效果,室温经过2 h反应能将低质量浓度甲醛(1.25 mg·m−3)降至0.018 mg·m−3以下。
2) UV-vis、PL、PEC和EIS表征结果显示,负载了CdS和Au纳米颗粒后的复合材料光吸收范围拓宽至800 nm,缺陷发光被抑制,促进了光生电子空穴分离和转移效率的提高,使其在可见光下仍保持较高的催化活性。
3) 测试结果显示催化剂种类、光源波长、相对湿度和甲醛质量浓度对甲醛去除效果影响较大,最佳光催化剂为ZnO/CdS/Au,在模拟太阳光 (300~800 nm) 照射下具有高甲醛去除效率,空气中湿度40%最宜,由于水分子会和甲醛分子竞争催化活性中心,2.5 mg·m−3作为反应起始质量浓度最佳,且催化剂具有循环稳定性。
制备ZnO/CdS/Au异质结用于可见光催化降解低浓度甲醛
Synthesis of ZnO/CdS/Au heterojunction for photocatalytic degradation of formaldehyde at room temperature under visible light
-
摘要: 采用水热法和化学浴沉积法制备了ZnO/CdS/Au异质结光催化剂,用于可见光下室温去除低浓度甲醛。通过XRD、SEM、TEM、XPS、UV、PL、PEC、EIS等手段对ZnO/CdS/Au异质结光催化剂的光学性能、光电性能进行表征。结果表明,Au负载的ZnO/CdS纳米棒组成的微米花状结构中,异质结和金属等离子体效应能拓宽光谱吸收范围,抑制半导体缺陷发光,促进光吸收以及光生电子空穴的分离和迁移;所制备的ZnO/CdS/Au异质结光催化剂对甲醛的去除具有优异性能,室温下2 h即可将反应舱内低质量浓度甲醛(1.25 mg·m−3)降至0.025 mg·m−3以下,且经过8次重复使用后催化剂活性没有明显下降。此外,影响甲醛去除的因素,如催化剂种类、光照波长、甲醛初始质量浓度、相对湿度也进行了研究和探讨。该研究结果对低浓度甲醛的室温去除具有理论指导意义。Abstract: ZnO/CdS/Au heterojunction photocatalysts were synthesized via hydrothermal and chemical bath deposition methods for the removal of low-concentration formaldehyde under visible light at room temperature. The optical and photoelectronic characteristics of the ZnO/CdS/Au heterojunction photocatalyst were systematically investigated utilizing a suite of analytical techniques, including XRD, SEM, TEM, XPS, UV, PL, PEC, EIS, and other complementary methods. The results revealed that the Au-decorated ZnO/CdS exhibited a micrometer-scale flower-like morphology composed of nanorods. The formation of the heterojunction and the plasmonic effect induced by the metal component led to an expansion of the spectral absorption range, suppression of the defect-related luminescence in the semiconductor, and enhancement of light absorption as well as efficient separation and migration of photogenerated electron-hole pairs. The fabricated ZnO/CdS/Au heterojunction photocatalyst demonstrated exceptional performance in the removal of formaldehyde, achieving the reduction of low-mass concentration formaldehyde (1.25 mg·m−3) in a reaction chamber to below 0.025 mg·m−3 within 2 h at room temperature. Moreover, the catalyst retained its activity with negligible degradation after 8 consecutive cycles. Furthermore, this study elucidated the influence of various factors on the removal of formaldehyde, such as the type of catalyst, light wavelength, initial mass concentration of formaldehyde, and relative humidity. A comprehensive discussion of these factors is presented herein. The findings of this research hold significant theoretical implications for the effective abatement of low-concentration formaldehyde at room temperature.
-
Key words:
- photocatalysis /
- heterojunction /
- metal /
- low concentration /
- formaldehyde
-

-
表 1 ZCAu中元素的绝对含量
Table 1. Absolute content for the elements of ZCAu
元素 元素含量/(mg·kg−1) 元素的质量分数/% Au 8 125.7 0.8 Cd 472 584.7 47.3 S 123 247.2 12.3 Zn 307 689.6 30.8 O — — 表 2 重复使用次数对ZCAu的HCHO去除效果及催化剂脱落的影响
Table 2. Effect of regeneration on HCHO removal and catalyst shedding for ZCAu
循环次数 反应后甲醛
质量浓度/ (mg·m−3)衬底和催化剂
质量/g1 0.018 16.75 2 0.029 16.74 3 0.039 16.71 4 0.046 16.66 5 0.049 16.62 6 0.041 16.58 7 0.051 16.55 8 0.065 16.48 -
[1] LEWIS A C, JENKINS D, WHITTY C J M. Hidden harms of indoor air pollution-five steps to expose them[J]. Nature, 2023, 614(7947): 220-223. doi: 10.1038/d41586-023-00287-8 [2] 王学川, 宋云云, 韩庆鑫. TiO2及其复合材料光催化降解室内甲醛的研究进展[J]. 功能材料, 2021, 52(5): 5076. doi: 10.3969/j.issn.1001-9731.2021.05.011 [3] 惠世恩, 朱新伟, 王登辉, 等. 活性炭负载TiO2吸附与光催化降解甲醛研究进展[J]. 洁净煤技术, 2022, 28(2): 1-12. [4] 杨振洲, 蔡同建. 室内甲醛的危害及其预防[J]. 中国公共卫生, 2003, 19(6): 765-768. doi: 10.3321/j.issn:1001-0580.2003.06.063 [5] CHEN X, GAO P, GUO L, et al. High-efficient physical adsorption and detection of formaldehyde using Sc-and Ti-decorated graphdiyne[J]. Physics Letters A, 2017, 381(9): 879-885. doi: 10.1016/j.physleta.2017.01.009 [6] ZHU X, JIN C, LI X S, et al. Photocatalytic formaldehyde oxidation over plasmonic Au/TiO2 under visible light: moisture indispensability and light enhancement[J]. ACS Catalysis, 2017, 7(10): 6514-6524. doi: 10.1021/acscatal.7b01658 [7] RAN M, CUI W, LI K, et al. Light-induced dynamic stability of oxygen vacancies in BiSbO4 for efficient photocatalytic formaldehyde degradation[J]. Energy & Environmental Materials, 2022, 5(1): 305-312. [8] 田景晨, 吴功德, 刘雁军, 等. 负载型廉价金属催化剂在低温催化氧化甲醛中的应用[J]. 化学进展, 2021, 33(11): 2069. [9] YE J, WANG L, ZHU B, et al. Light-enhanced metal-support interaction for synergetic photo/thermal catalytic formaldehyde oxidation[J]. Journal of Materials Science & Technology, 2023, 167: 74-81. [10] TASBIHI M, BENDYNA J K, NOTTEN P H L. A short review on photocatalytic degradation of formaldehyde[J]. Journal of nanoscience and nanotechnology, 2015, 15(9): 6386-6396. doi: 10.1166/jnn.2015.10872 [11] NEMIWAL M, ZHANG T C, KUMAR D. Recent progress in g-C3N4, TiO2 and ZnO based photocatalysts for dye degradation: Strategies to improve photocatalytic activity[J]. Science of the total environment, 2021, 767: 144896. doi: 10.1016/j.scitotenv.2020.144896 [12] ZHANG Y, QIU J, ZHU B, ET AL. ZnO/COF S-scheme heterojunction for improved photocatalytic H2O2 production performance[J]. Chemical Engineering Journal, 2022, 444: 136584. doi: 10.1016/j.cej.2022.136584 [13] GOKTAS S, GOKTAS A. A comparative study on recent progress in efficient ZnO based nanocomposite and heterojunction photocatalysts: A review[J]. Journal of Alloys and Compounds, 2021, 863: 158734. doi: 10.1016/j.jallcom.2021.158734 [14] WIBOWO A, MARSUDI M A, AMAL M I, et al. ZnO nanostructured materials for emerging solar cell applications[J]. RSC advances, 2020, 10(70): 42838-42859. doi: 10.1039/D0RA07689A [15] WANG Y, ZHENG Z, WANG J, et al. New Method for Preparing ZnO Layer for Efficient and Stable Organic Solar Cells[J]. Advanced Materials, 2023, 35(5): 2208305. doi: 10.1002/adma.202208305 [16] SIVASAKTHI S, GURUNATHAN K. Graphitic carbon nitride bedecked with CuO/ZnO hetero-interface microflower towards high photocatalytic performance[J]. Renewable Energy, 2020, 159: 786-800. doi: 10.1016/j.renene.2020.06.027 [17] LAHMAR H, BENAMIRA M, DOUAFER S, et al. Photocatalytic degradation of methyl orange on the novel hetero-system La2NiO4/ZnO under solar light[J]. Chemical Physics Letters, 2020, 742: 137132. doi: 10.1016/j.cplett.2020.137132 [18] HADDAD M, BELHADI A, BOUDJELLAL L, et al. Photocatalytic hydrogen production on the hetero-junction CuO/ZnO[J]. International Journal of Hydrogen Energy, 2021, 46(75): 37556-37563. doi: 10.1016/j.ijhydene.2020.11.053 [19] ZAMAN F, XIE B, ZHANG J, et al. MOFs derived hetero-ZnO/Fe2O3 nanoflowers with enhanced photocatalytic performance towards efficient degradation of organic dyes[J]. Nanomaterials, 2021, 11(12): 3239. doi: 10.3390/nano11123239 [20] NEENA D, HUMAYUN M, ZUO W, et al. Hierarchical hetero-architectures of in-situ g-C3N4-coupled Fe-doped ZnO micro-flowers with enhanced visible-light photocatalytic activities[J]. Applied Surface Science, 2020, 506: 145017. doi: 10.1016/j.apsusc.2019.145017 [21] SHEN R, REN D, DING Y, et al. Nanostructured CdS for efficient photocatalytic H2 evolution: A review[J]. Science China Materials, 2020, 63(11): 2153-2188. doi: 10.1007/s40843-020-1456-x [22] SUN G, XIAO B, ZHENG H, et al. Ascorbic acid functionalized CdS-ZnO core-shell nanorods with hydrogen spillover for greatly enhanced photocatalytic H2 evolution and outstanding photostability[J]. Journal of Materials Chemistry A, 2021, 9(15): 9735-9744. doi: 10.1039/D1TA01089A [23] LU H, LIU Y, ZHANG S, et al. Clustered tubular S-scheme ZnO/CdS heterojunctions for enhanced photocatalytic hydrogen production[J]. Materials Science and Engineering:B, 2023, 289: 116282. doi: 10.1016/j.mseb.2023.116282 [24] 郭俊兰, 梁英华, 王欢, 等. 光催化制氢的助催化剂[J]. 化学进展, 2020, 33(7): 1100. doi: 10.7536/PC200803 [25] AHMAD M, REHMAN W, KHAN M M, et al. Phytogenic fabrication of ZnO and gold decorated ZnO nanoparticles for photocatalytic degradation of Rhodamine B[J]. Journal of Environmental Chemical Engineering, 2021, 9(1): 104725. doi: 10.1016/j.jece.2020.104725 [26] SANAKOUSAR F M, VIDYASAGAR C C, JIMÉNEZ-PÉREZ V M, et al. Recent progress on visible-light-driven metal and non-metal doped ZnO nanostructures for photocatalytic degradation of organic pollutants[J]. Materials science in semiconductor processing, 2022, 140: 106390. doi: 10.1016/j.mssp.2021.106390 [27] KARTHIK K V, RAGHU A V, REDDY K R, et al. Green synthesis of Cu-doped ZnO nanoparticles and its application for the photocatalytic degradation of hazardous organic pollutants[J]. Chemosphere, 2022, 287: 132081. doi: 10.1016/j.chemosphere.2021.132081 [28] LI X S, MA X Y, LIU J L, et al. Plasma-promoted Au/TiO2 nanocatalysts for photocatalytic formaldehyde oxidation under visible-light irradiation[J]. Catalysis Today, 2019, 337: 132-138. doi: 10.1016/j.cattod.2019.03.033 [29] VEZIROGLU S, OBERMANN A L, ULLRICH M, et al. Photodeposition of Au nanoclusters for enhanced photocatalytic dye degradation over TiO2 thin film[J]. ACS Applied Materials & Interfaces, 2020, 12(13): 14983-14992. [30] YU Z B, XIE Y P, LIU G, et al. Self-assembled CdS/Au/ZnO heterostructure induced by surface polar charges for efficient photocatalytic hydrogen evolution[J]. Journal of Materials Chemistry A, 2013, 1(8): 2773-2776. doi: 10.1039/c3ta01476b [31] ZHAO X, WU Y, HAO X. Electrodeposition synthesis of Au-ZnO hybrid nanowires and their photocatalytic properties[J]. International Journal of Electrochemical Science, 2013, 8(3): 3349-3356. doi: 10.1016/S1452-3981(23)14395-1 [32] ZHANG N, XIE S, WENG B, et al. Vertically aligned ZnO-Au@CdS core-shell nanorod arrays as an all-solid-state vectorial Z-scheme system for photocatalytic application[J]. Journal of Materials Chemistry A, 2016, 4(48): 18804-18814. doi: 10.1039/C6TA07845A [33] JIMENEZ-SALCEDO M, MONGE M, TERESA TENA M. An organometallic approach for the preparation of Au-TiO2 and Au-g-C3N4 nanohybrids: Improving the depletion of paracetamol under visible light[J]. Photochemical & Photobiological Sciences, 2022, 21(3): 337-347. [34] WEI R B, KUANG P Y, CHENG H, et al. Plasmon-enhanced photoelectrochemical water splitting on gold nanoparticle decorated ZnO/CdS nanotube arrays[J]. ACS Sustainable Chemistry & Engineering, 2017, 5(5): 4249-4257. [35] SHAFIQ F, TAHIR M B, HUSSAIN A, et al. The construction of a highly efficient p-n heterojunction Bi2O3/BiVO4 for hydrogen evolution through solar water splitting[J]. International Journal of Hydrogen Energy, 2022, 47(7): 4594-4600. doi: 10.1016/j.ijhydene.2021.11.075 [36] FU H, MA Y, YANG Z, et al. Construction of MnFe2O4/Bi5O7I composite heterojunction and its visible light-driven photocatalytic degradation of RhB[J]. Ionics, 2022, 28(8): 3893-3905. doi: 10.1007/s11581-022-04626-z [37] JIANG T, WANG X, CHEN J, et al. Hierarchical Ni/Co-LDHs catalyst for catalytic oxidation of indoor formaldehyde at ambient temperature[J]. Journal of Materials Science:Materials in Electronics, 2020, 31: 3500-3509. doi: 10.1007/s10854-020-02898-7 -




 下载:
下载: