-
全氟化合物(perfluorinated compounds, PFCs)是消防泡沫、石油勘探、食品包装、服装和纺织品等生产活动的常见排放物。近年来,由于我国氟化工产业迅速发展造成其在多种水体中频繁检出[1-3]。SUN等[4]调查了上海市39个地表水样品中17种PFCs的污染状况,发现有10种PFCs检出。QI等[5]在天津市地下水中检测到13种全氟辛烷磺酸,其中全氟辛酸(perfluorooctanoic acid, PFOA)和全氟丁烷磺酸(perfluorobutane sulfonic acid, PFBS)含量最高。LI等[6]调研了中国79个城市饮用水中17种PFCs的含量,总浓度为4.49~174.93 ng·L−1,平均值为35.13 ng·L−1。PFCs通过呼吸、饮水和饮食等途径进入人体后,对肝脏功能、脂肪代谢和遗传发育均有不良影响,可能导致哮喘,肝肿瘤和慢性肾病等[7]。因此,亟需研发水中PFCs的高效去除技术。
PFCs分子具有高能C—F键,其结构性质稳定且极难被氧化或微生物降解。吸附是目前去除水PFCs最为经济可行的技术,常用的吸附剂主要有活性炭、树脂等。其中活性炭作为一种具有发达孔隙结构及高比表面积的广谱吸附剂,因其相对低廉的成本而被广泛应用。大量研究表明活性炭在去除PFCs方面,尤其是长链PFCs,具有较好的应用潜能。例如,孙博等[8]发现采用椰壳粉末炭实现了超纯水中90%以上长链PFOA的去除,但对于短链全氟丁酸(perfluorobutyric acid, PFBA)去除效果不佳(低于10%)。SON等[9]研究也发现煤质和椰壳粉末活性炭对长链全氟辛烷磺酸(perfluorooctanesulfonic acid, PFOS)和PFOA去除率分别可达50%~95%和30%~90%,但对短链PFBS去除率仅为20%~40%。因此,活性炭吸附是目前去除水中PFCs最为可行处理技术工艺,然而如何将活性炭吸附与其他工艺组合提升PFCs,尤其是短链PFCs的处理效能,仍需进一步深入研究。
在目前的净水工艺中,尤其在农村净水工艺中,普遍采用高级氧化与吸附技术相结合。基于UV的高级氧化技术操作简单、氧化能力强、且二次污染较小,得到了广泛应用。该过程主要通过光化学反应激发氧化剂产生高反应活性的自由基实现污染物的高效去除,其中UV与H2O2的组合是最常见的高级氧化过程。JAVED等[10]的研究表明在UV体系中H2O2对水中PFOA去除率的提高没有明显作用,UV的直接光降解是PFOA的主要降解机制。然而TANG等[11]的研究得出了不同的结果,报道显示UV-Fenton高级氧化在1h内可实现87.9%的PFOA去除率,且UV照射时长和氧化剂质量浓度对PFOA的去除有一定影响,氧化剂质量浓度增加对PFOA的去除有一定增益,但超过20 mmol·L−1时,PFOA的去除率会降低。此外,PFCs的初始质量浓度对高级氧化对其的降解效率有一定影响,质量浓度较高时PFCs降解速率受到抑制,因此研究人员建议PFCs的高级氧化降解探究应采用μg·L−1质量浓度水平[12]。目前关于高级氧化降解PFCs的研究主要集中在PFOA和PFOS上,PFOS可以经自由基反应转化为PFOA,最后转化为氟离子、甲酸和二氧化碳等[13-14]。虽然对于高级氧化技术去除PFCs有了一定的研究进展,然而高价氧化处理污染水体,会存在矿化不完全的问题,易形成毒性较高的中间产物,因此需要与活性炭吸附技术联用,消除中间产物的潜在环境风险。此外,目前对于高级氧化与活性炭吸附联合处理PFCs的报道较少,且已有研究存在一定的分歧,因此针对水中PFCs去除的UV高级氧化与吸附联用处理技术的进一步探究是有必要的。
因此,本研究利用快速小柱实验法(rapid small-scale column tests, RSSCT),对比分析了UV/GAC、H2O2/GAC和UV/H2O2/GAC 3种组合工艺对水中不同链长的PFCs包括PFBA、PFBS、PFOA和PFOS的去除效果和影响机制,以期为水中PFCs去除的工艺选择提供科学依据。
-
实验用水是在西北黄土高原地区偏远乡镇水窖水中加入PFBA、PFBS、PFOA和PFOS混合2 h得到的。为了避免提取和浓缩过程的偏差,实验水中4种PFCs的质量浓度分别达到200 μg·L−1。原水的水质参数如下:浊度为0.74 NTU;pH =7.76;UV254为0.038;TOC为2.609 mg·L−1;总溶解固体为110.0 mg·L−1;溶解氧为8.8 mg·L−1;Zeta电位为-7.47~-11.2 mV;氧化还原电位为250.5 mV。
-
亚硫酸钠(Na2SO3,国药集团化学药剂有限公司)、过氧化氢(H2O2,30%,北化)、盐酸、氢氧化钠;颗粒活性炭(椰壳50~100目,碘值1 050 mg·g−1,强度97%,用抽滤装置洗至pH为7.0±0.2;紫外灯(254 nm):10 W,平均光强1.87 mW·cm−2(通过阿特拉津化学辐射剂量法测定);全氟丁酸(PFBA)、全氟丁烷磺酸(PFBS)、全氟辛酸(PFOA)、全氟辛烷磺酸(PFOS)储备液质量浓度100 mg·L−1。为了避免富集前处理过程产生的测量误差,实验中使各反应器中4种PFCs的初始质量浓度均为200 μg·L−1。4种PFCs储备液加入500 mL窖水烧杯中,通过磁力搅拌器,搅拌2 h实现PFCs混合均匀。UV/H2O2/GAC工艺去除水中4种PFCs的探究中,H2O2的质量浓度均为10 mg·L−1。
-
分析天平;pH计(FE28,梅克托,瑞士);磁力搅拌器;蠕动泵(兰格,中国河北);紫外-可见分光光度仪(MAPADA,中国上海);三重四极杆液相色谱质谱联用仪(安捷伦6460,美国)。
-
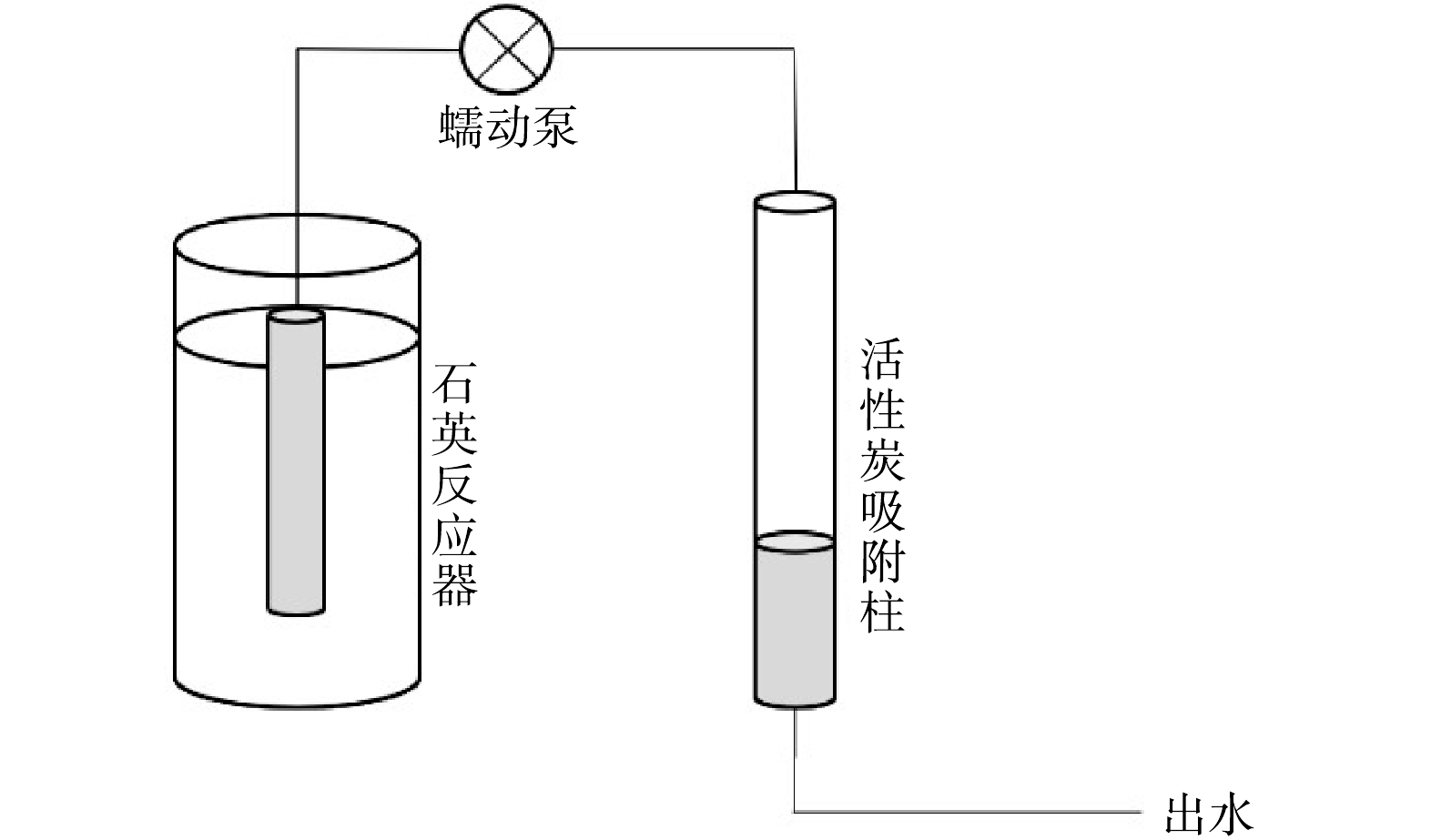
UV定制透明石英圆柱形水桶直径100 mm、高度200 mm、孔径8 mm,出水口管道30 mm。为保证UV辐射输出保持稳定状态,实验前对UV灯预热10 min,实验在(25±2) °C下进行。实验装置示意见图1。
-
小型化快速柱式反应实验(rapid small scale column tests, RSSCT)被广泛应用于实验室中,可以准确预测中试规模的大柱吸附污染物的过程,具有水量需求小,实验时间短等优点。本章根据PD-RSSCT设计法[15](式(1))。
其中:Ex和Ed分别表示设计小柱和大柱的空床接触时间,min;Rx和Rd分别表示设计小柱和大柱的半径,cm;tx和td分别表示设计小柱和大柱的运行时间,min。
采用高20 cm,内径2 cm的层析柱作为小柱子,填充100~50目(150~300 μm)的GAC。GAC填充高度为4 cm,内径2 cm,空床体积12.6 cm3,设计空床接触时间(EBCT)为2.5 min,进水流量5 mL·min−1。
模拟大柱设计流量为3 000 mL·min−1,模拟GAC粒度24~12目(
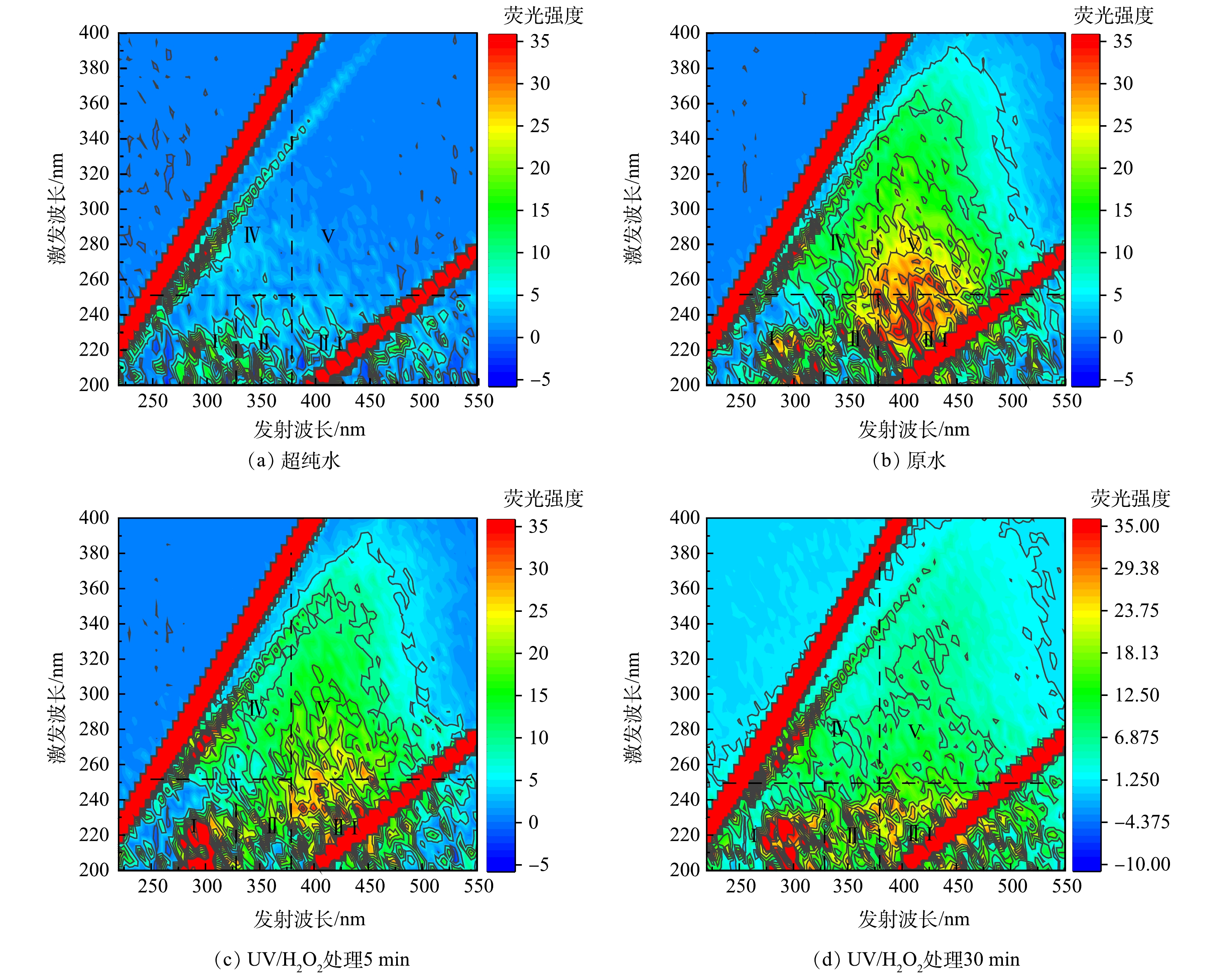
$ 710~1\mathrm{ }\mathrm{ }\mathrm{ }\mathrm{ }400\mathrm{ }\mathrm{\mu }\mathrm{m} $ ),设计空床接触时间(empty bed contact time, EBCT)为10 min,大柱滤床体积30 L,直径0.37 m,高0.56 m。UV/H2O2和UV/H2O2/GAC 2个体系的出水经0.45 μm滤膜过滤后进行三维荧光的检测,利用matlab对三维荧光光谱进行积分,得到各区组分含量。三维荧光光谱表示的5类物质[16]如表1所示。
出水4种PFCs样品中的4种PFCs采用外标法在电喷雾离子源负离子模式(ESI-)下用美国安捷伦公司1290 Infinity HPLC系统与Agilent 6460三重四极杆质谱系统联用的高效液相色谱串联质谱仪进行检测。气源温度350 ℃;气源流速10 mL·min−1;雾化器压力35 psi;毛细管电压3 500 V。检测中使用的色谱柱为ZORBAX RR Eclipse Plus C18, 2.1 mm×100 mm, 3.5 μm (Agilent, USA);柱温40 ℃;进样量5 μL;流速0.4 mL·min−1;洗脱程序:0~0.3 min内乙腈:超纯水=30%:70%,0.5~2.5 min内乙腈:超纯水=90%:10%, 3.6~6.0 min内乙腈:超纯水=30%:70%,4种PFCs的质谱分析条件如表2所示。
-
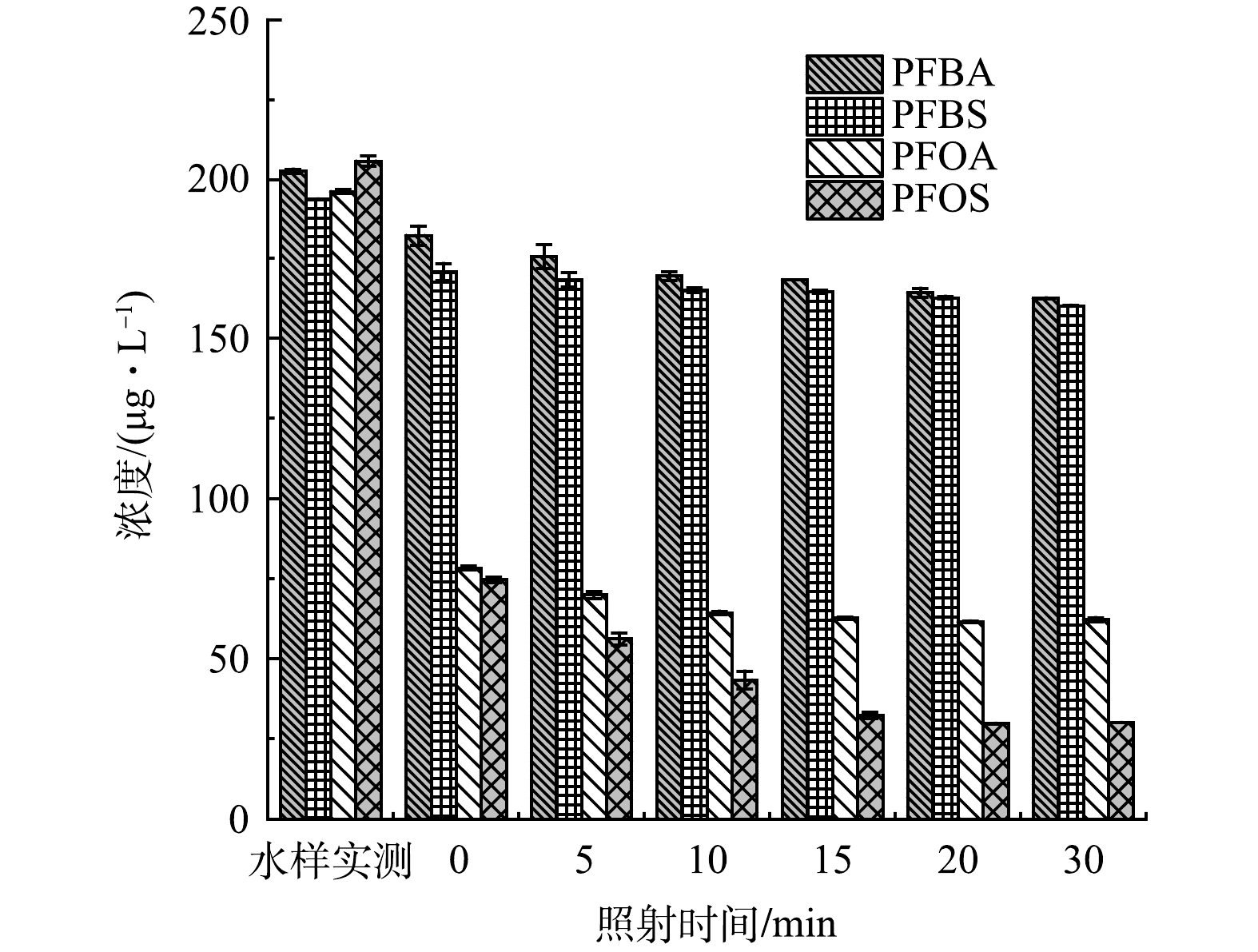
不同UV照射时长(0、5、10、15、20、30 min)对于GAC吸附水中4种PFCs效果的影响如图2所示。水样实测表示的是不经任何处理的4种PFCs实测值,0表示不经UV照射仅进行GAC吸附处理,横坐标其他数值表示经相应时间UV照射再进行GAC吸附处理。结果表明,仅进行GAC吸附时,PFBA和PFBS去除率分别为11.7%和13.1%。随着UV照射时间的增加,PFBA和PFBS去除率均有一定的增加,最高去除率分别达到19.8%和17.1%。相比PFBA和PFBS,PFOA和PFOS具有更显著的去除效果,仅GAC单独吸附就可达到59.6%和64.3%。UV照射20 min后,PFOA和PFOS去除率分别增加至68.3%和85.7%。PFCs的去除主要受疏水性的影响,而官能团种类和链长是影响其在水中疏水性的重要原因。对于PFOA和PFOS,其分子中均含有8个碳原子较分子中含有4个碳原子的PFBA和PFBS有更长的链长,疏水性更强,去除率更高[17]。此外,虽然PFOA和PFOS有相同的C-F链长度,但实验中带有磺酸基团的PFOS去除率始终高于带有羧酸基团的PFOA,表明活性炭上的吸附位点更容易被磺酸基团占据。羧酸基团中的C=O键易于受到质子化的影响具有更强的共振效应,基团中的羟基更易于失去氢而使整个羧基成为羧酸根阴离子,具有更强的酸性和亲水性。而磺酸基团中的S=O键极性大,结构更稳定不易发生电离,疏水性相对于羧酸基团更强[18]。总体来说,254 nmUV灯照射一定程度上可以增益GAC去除水中4种PFCs的效果,但增益效果较弱。
根据已有研究中单独UV降解PFCs的报道结果,在超纯水、自来水、河水和污水处理厂出水等多种不同背景水质条件下均显示单独UV无法有效去除PFCs,如CAO等[19]用254 nm的紫外灯降解水溶液中的PFOA,发现照射2 h后仅观察到9%的PFOA降解,只有与其他处理技术联用才会取得良好的PFCs去除效果[20-21]。光解效率取决于化合物吸收发射光的能力和紫外光对溶液的透过率,254 nm属于低压汞灯的发射波长,PFCs在190~280 nm紫外照射区域吸收率非常低,降解效果不明显,而在200 nm以下的真空紫外照射区域有更显著的吸收且对溶液的透过率有所增加,对PFCs的降解效果明显增强,185 nm紫外灯照射2 h后,PFOA去除率和脱氟率分别达到79%和18%[22]。然而,真空紫外的经济性和安全性是在实际应用中要考虑的重要问题。此外,相关研究表明,PFCs的去除效果与水中天然有机物的存在形态和含量有一定关系,紫外光照射下一定程度上改变了天然有机物的化学和生物性质,天然有机物的存在可能影响紫外光对反应溶液的透过率,有研究表明,在大量有机物基质存在的情况下用于PFCs光降解的能量比例急剧下降[23-24]。
-
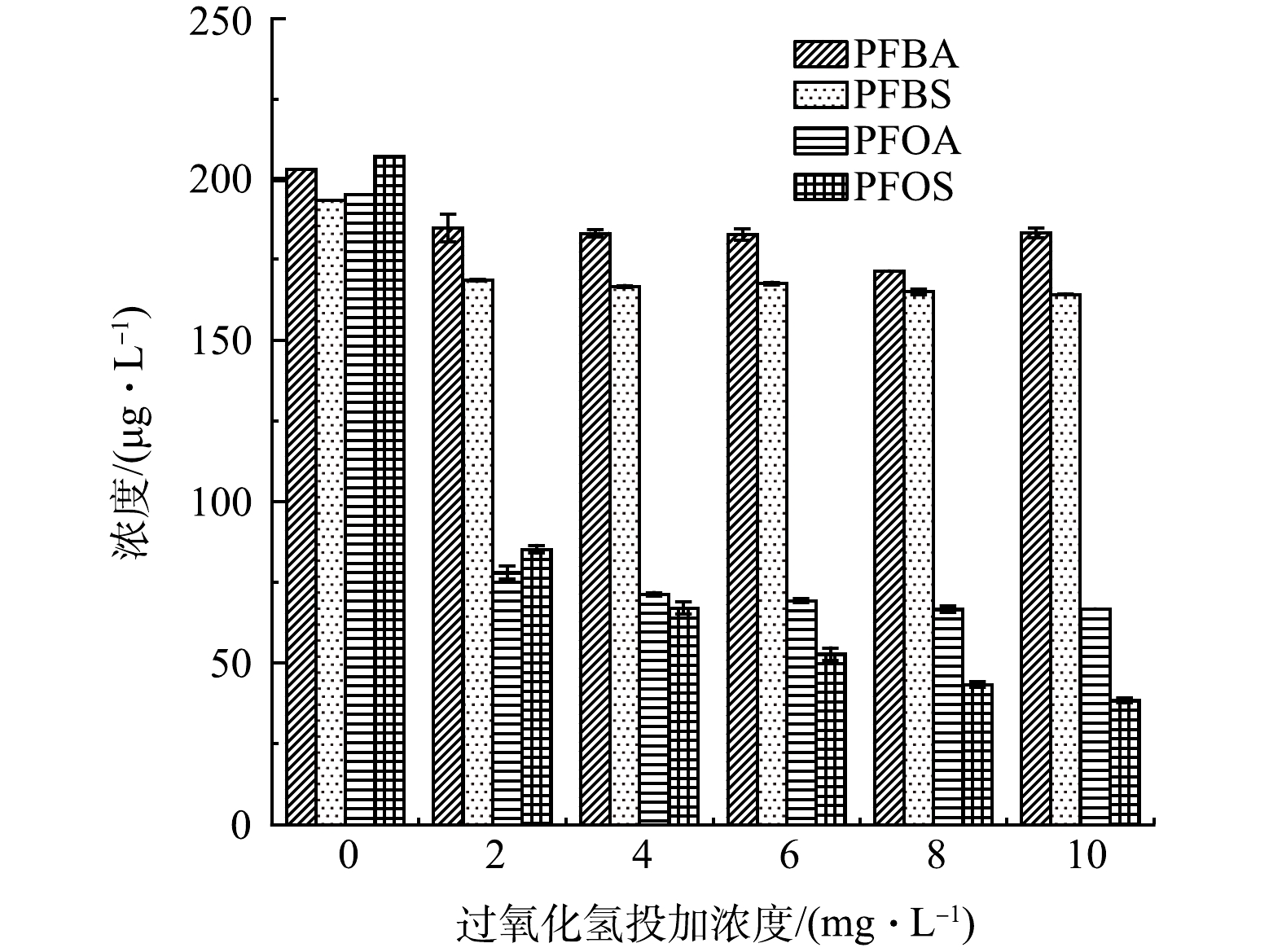
在考察了UV光直接降解与GAC联用去除水中4种PFCs后,本节探讨了H2O2直接氧化与GAC联用对4种PFCs的去除效果,结果如图3所示。H2O2质量浓度增加对PFBA、PFBS、PFOA和PFOS的去除均无明显增强效果,甚至在高质量浓度下会降低PFCs的去除效果,这可能与H2O2对水中天然有机物的去除有关。高质量浓度H2O2下体系中羟基自由基的产生量反而有所降低,有研究表明,腐殖酸的H2O2最佳降解浓度为0.003 2~0.016 3 mol·L−1 [25]。本实验中,水样经2 mg·L−1 H2O2处理后,PFBS、PFOA和PFOS的去除率从13.1%、59.6%和64.3%分别降为12.9%、59%和59.4%,H2O2直接氧化较254 nm UV直接光降解对PFCs的去除效果更差,这可能是由于两者对PFCs攻击位置不同所致。根据光子能量值和平均键能,PFOA中的C—C键的键能为347.0 kJ·mol−1,而C—F键的键能为552.0 kJ·mol−1,相对于C—C键,C—F难以作为UV攻击位点而被断裂,因此UV作用的是PFCs分子中的C—C键[26]。而对于PFCs的氧化来说,由于F原子周围高电子云密度强屏蔽效应钝化了活性氧诱导的氧化过程,氧化效率相对较低[27]。此外,有研究[28]表明,仅用紫外照射或者H2O2直接氧化时水中天然有机物去除效果也并不显著。然而,与H2O2直接氧化不同,直接光降解对UV254指标有一定的降低。UVA照射时的最大吸光度损失为270~410 nm、UVB照射时吸光度损失为260~370 nm和UVC照射时为230~300 nm。水中天然有机物对UVC的照射损失明显高于UVA和UVB[29]。因此,紫外光降解实现了一些大分子有机物向小分子有机物的转化,对PFCs的去除有一定促进作用。相反,当使用过高浓度的H2O2时,可能会对羟基自由基产生清除作用,反而会降低体系中PFCs的去除效果。
-
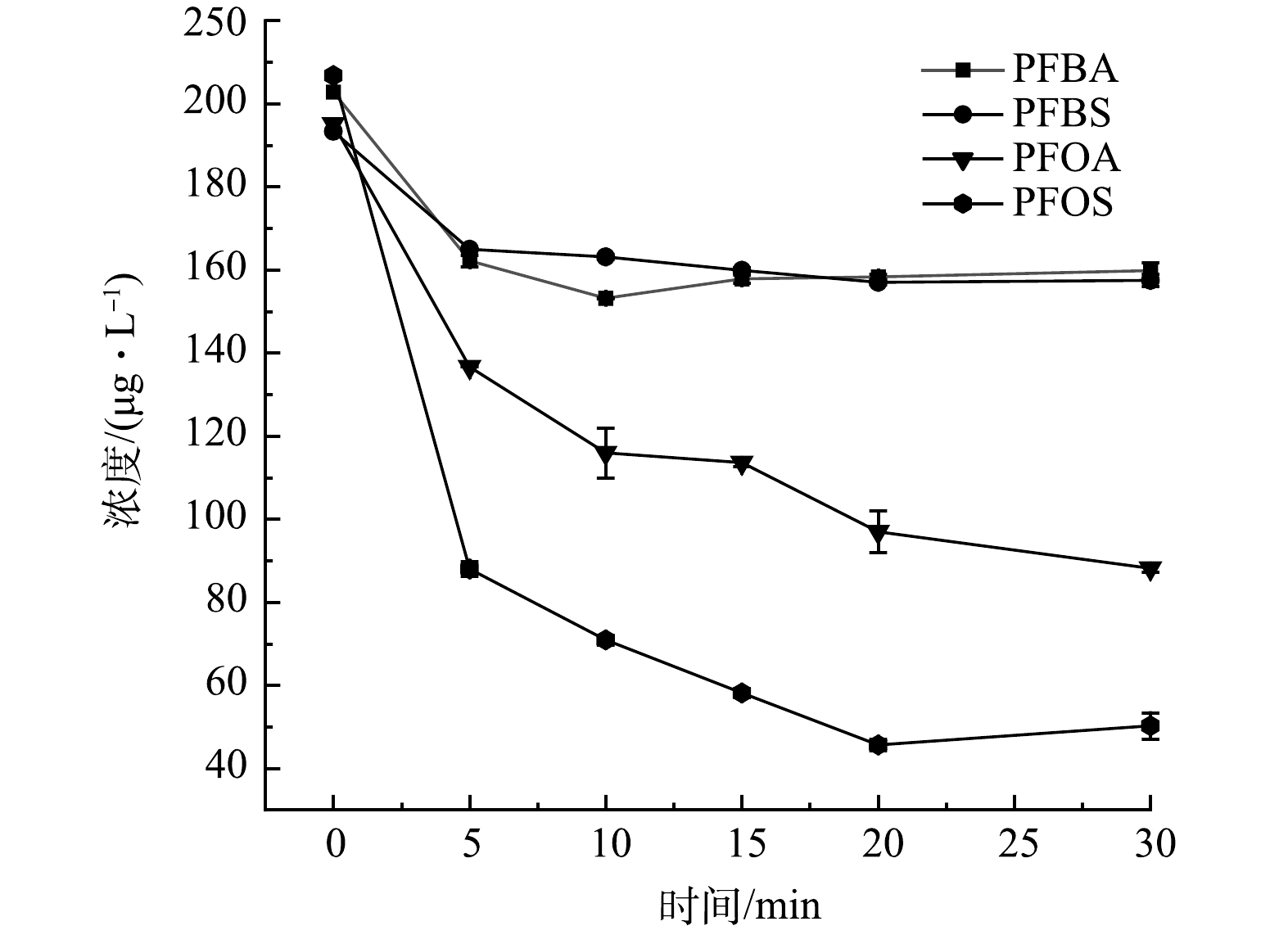
首先考察了仅经过UV/H2O2处理,不采用GAC吸附对水中4种PFCs的去除效果,实验中H2O2投加量均为10 mg·L−1。如图4所示,在0~30 min内,PFBA的去除率为19.3%~24.4%,PFBS的去除率为14.6%~19.1%,PFOA的去除率为30%~55.3%,PFOS的去除率为56.6%~77.3%。PFBA和PFBS去除率在10 min内达到平台期,10~30 min内去除率未有明显增加,PFOA和PFOS去除率在0~20 min内均有一定的增加的趋势。有研究表明,对于PFOA的降解来说,无论基质差异和有效辐照剂量如何变化,直接紫外光降解是PFOA降解的主要机制,而羟基自由基贡献不大。在0、0.5 和5 g·L−1的H2O2质量浓度条件下,PFOA的降解效果均无明显差异,且过量的H2O2会阻碍紫外光的渗透进而影响PFOA的降解。这表明不经过GAC吸附时,UV/H2O2对PFCs的去除主要是UV照射贡献。然而,UV/H2O2对水中天然有机物的降解被证明是有效的,且随着投加H2O2质量浓度的增加,水中有机物的粒径逐渐减小,在紫外线照射120 min后,分子质量大于30 kDa的腐植酸类产物明显减少,其浓度降低了90%。另一方面,在120 min时体系中天然有机物粒径基本小于10 kDa。由此可见,UV/H2O2基础上引入GAC吸附可强化PFCs的去除效果[25]。
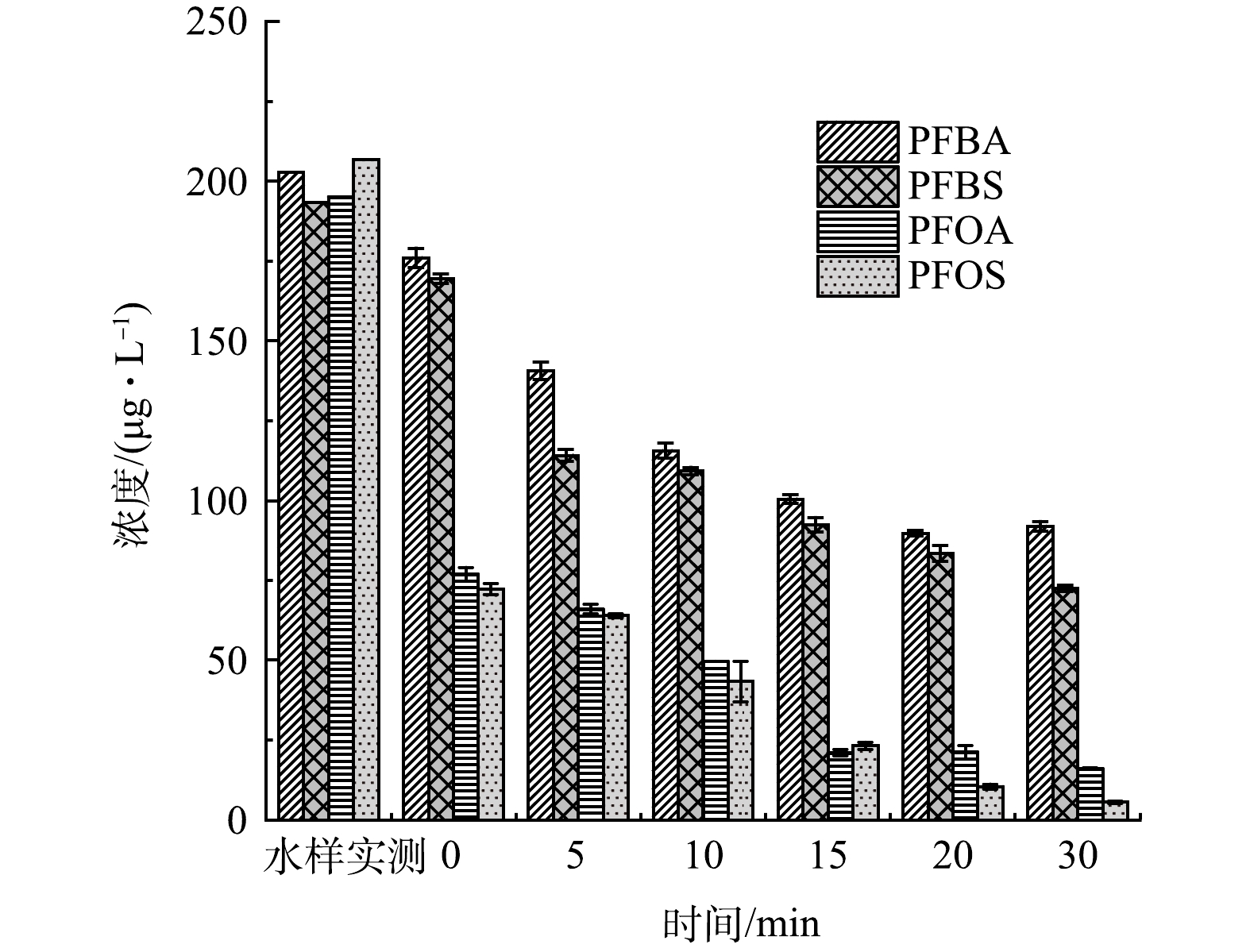
将UV/H2O2出水泵入GAC吸附柱,进一步探究了UV/H2O2/GAC对水中4种PFCs的去除效果。如图5所示,经UV/H2O2处理20 min后,GAC吸附柱出水中PFBA去除率达到最高55.3%;UV/H2O2处理30 min后GAC吸附柱出水中PFBS、PFOA和PFOS去除率达到最高为60%、91.6%和97.4%。相比于仅UV处理后经过GAC吸附柱出水,PFBA、PFBS、PFOA、PFOS的去除率分别提高了35.5%、42.9%、23.3%、11.7%。相比于仅经H2O2处理后再经过GAC吸附柱出水,PFBA、PFBS、PFOA、PFOS的去除率分别提高了42.8%、46.9%、32%、16.3%。
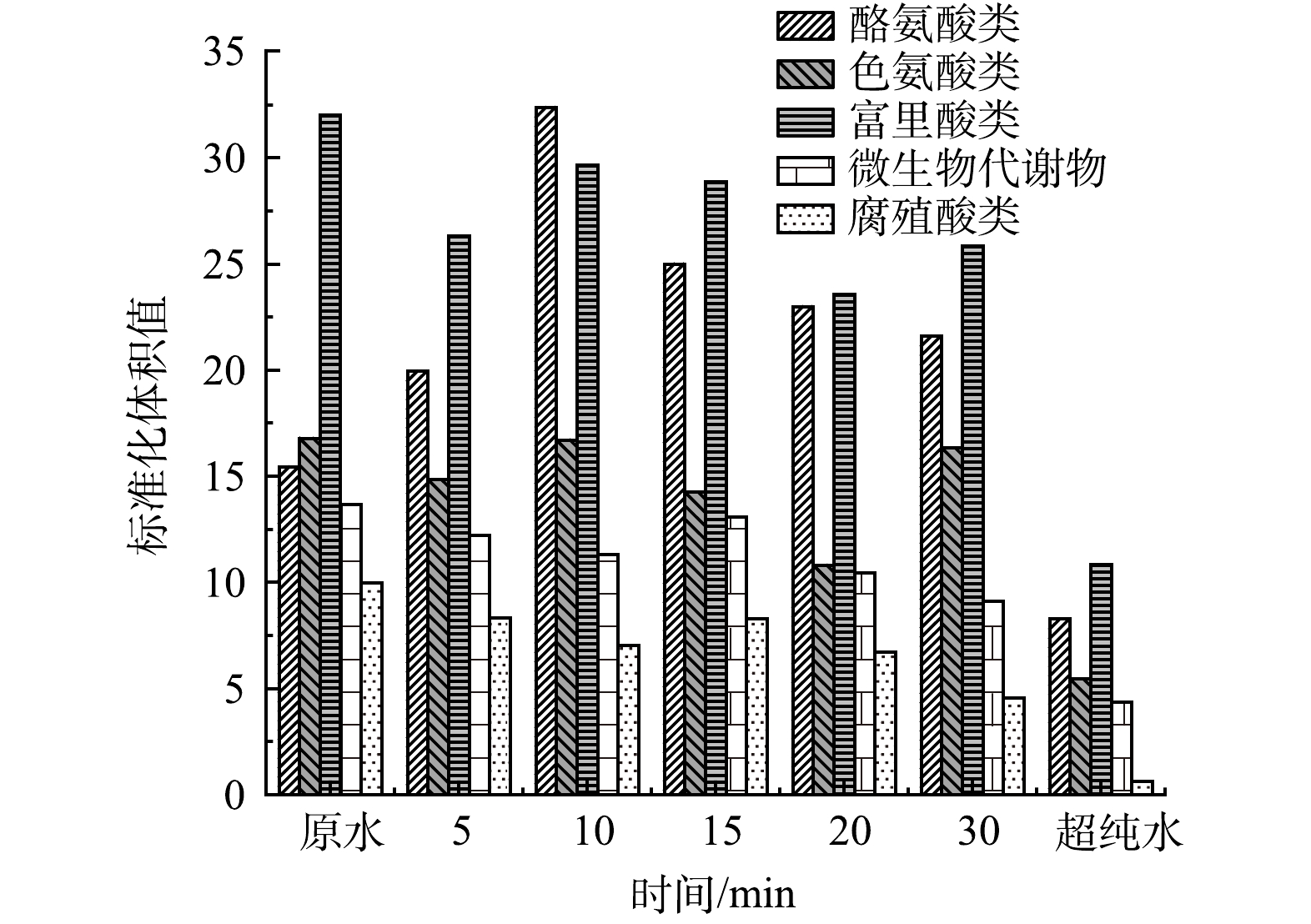
鉴于单独UV/H2O2对于水中PFCs去除效果并不明显,推测UV/H2O2/GAC对PFCs较好的去除效果是由于UV/H2O2/高级氧化体系改变了水中有机基质的结构和形态,从而有利于GAC的吸附。其中PFBA和PFBS去除率平台期的出现很可能是与两种化合物结合的大分子有机物经过了充分降解后达到了相对稳定的状态。因此,对UV/H2O2处理后的水样进行了三维荧光检测,并利用matlab进行了区域积分,量化了高级氧化前后天然有机物组成和种类的变化。图6为UV/H2O2前后三维荧光光谱图。可以看到,相比于超纯水的三维荧光光谱,在加药原水中,代表酪氨酸类、富里酸类和腐殖酸类的Ⅰ、Ⅲ和Ⅴ区荧光强度较高。UV/H2O2处理5 min和30 min后,Ⅰ、Ⅲ和Ⅴ区荧光强度明显降低。对各区域积分标准化得到了各部分的表征含量,如图7所示,随着UV/H2O2处理时长的增加,除酪氨酸增加39.8%外,色氨酸、富里酸、腐殖酸和微生物产物的标准化体积去除率均有下降趋势,在处理30 min后去除率分别达到27.3%、19.3%、54.5%和33.4%。
水中的天然有机物是活性炭吸附PFCs的主要竞争者且天然有机物的形态大小对PFCs在活性炭上的吸附有显著影响。大分子有机物具有很强的疏水性,易于吸附在GAC上与PFCs形成竞争吸附,GAC上的吸附位点大大减少,产生空间位阻效应,从而影响了PFCs在GAC上的吸附。有研究[30]表明,有机物质量浓度较高时,有机物大分子的空间位阻效应以及竞争吸附作用会使PFOA和PFOS的吸附速率降低20%~44%。此外,腐殖酸等天然有机物的羧酸类基团可以在水中解离而使整个有机物分子带有负电,这使得天然有机物与同样带负电的PFCs产生静电斥力,天然有机物吸附在颗粒活性炭上后阻止了PFCs的进一步吸附。经过高级氧化处理后,大分子有机物被氧化成分子质量更小的有机物,因而疏水性下降,且不会导致孔结构堵塞,更利于GAC吸附PFCs,因此,UV/H2O2对GAC吸附PFCs具有一定的增强效果[31-35]。
GAC相对粉末活性炭(PAC)来说成本并不高,具有一定经济性,而UV/H2O2/GAC联用对水中4种PFCs的削减效果最强,因此,相比UV/GAC、H2O2/GAC和UV/H2O2,UV/H2O2/GAC是更佳的组合处理工艺。
-
1) UV/GAC联用去除水中4种PFCs时,PFBA、PFBS、PFOA和PFOS去除率随着UV照射时间的延长略有增加,最高去除率分别为19.8%、17.1%、68.3%和85.7%。链长较PFBA和PFBS更长的PFOA和PFOS更易于在体系中去除,具有磺酸基的PFOS相较于羧酸基的PFOA更易于在体系中去除。
2) H2O2/GAC联用去除水中4种PFCs时,H2O2质量浓度增加对PFBA、PFBS、PFOA和PFOS的去除效果均无明显的影响,甚至在高质量浓度下PFCs的去除效果有所降低。经2 mg·L−1 H2O2处理后,PFBS、PFOA和PFOS的去除率由13.1%、59.6%和64.3%分别降为12.9%、59%和59.4%。
3) UV/H2O2产生的羟基自由基使水中大分子有机物含量降低,色氨酸、富里酸、腐殖酸和微生物产物的标准化体积均有下降趋势,经过30 min处理,各组分分别下降了27.3%,19.3%、54.5%和33.4%。
4) UV/H2O2/GAC联用对4种PFCs的去除效果最好,仅需30 min UV/H2O2处理,经过GAC吸附柱的出水中有50%以上的PFBA和PFBS得以去除,90%以上的PFOA和PFOS得以去除,这表明该方法对PFCs处理具有较好的应用前景。
UV/H2O2/GAC工艺去除水中全氟化合物
Removal of PFCs from water by UV/ H2O2/GAC process
-
摘要: 全氟化合物(PFCs)持久存在于水环境中,难以通过传统的水处理工艺去除,提高水中PFCs的去除性能具有重要意义。本研究采用快速小柱实验探究了UV、H2O2和颗粒活性炭(GAC)组合工艺对水中4种PFCs的去除效果,并探究了UV照射时长和H2O2质量浓度对PFCs的直接去除效果及其对后续GAC吸附的影响。结果表明:不同工艺条件下,长链全氟辛酸(PFOA)和全氟辛烷磺酸(PFOS)始终表现出较短链全氟丁酸(PFBA)和全氟丁烷磺酸(PFBS)更高的去除率。单独GAC吸附对长链全氟PFOA和PFOS去除率为59.6%和64.3%,但对短链PFBA和PFBS去除率仅为11.7%和13.1%。单独UV或单独H2O2分别与GAC联用时,随着UV照射时长和H2O2质量浓度的增加,4种PFCs的去除率略有增加,且UV照射的增益效果较H2O2更优。UV/H2O2/GAC联用工艺对水中4种PFCs的去除效果最佳,30 min UV/H2O2处理后经GAC吸附可去除90%以上的PFOA和PFOS,短链PFBA和PFBS去除率也达到50%以上。UV/H2O2/GAC联工艺的良好效果是由于UV/H2O2高级氧化过程产生的羟基自由基氧化反应降低了水中大分子有机物的含量,从而减弱了有机物对PFCs的竞争吸附作用。本研究可为水中PFCs的高效处理提供技术参考。Abstract: Perfluorinated compounds (PFCs) are persistently found in aquatic environment, which are difficult to be removed by the conventional water treatment process. Improving the removal effect of PFCs in water is of great significance. The removal efficacies of four kinds of PFCs in water by the combined process of UV, H2O2 and GAC were studied using the rapid small-scale column tests (RSSCT), and the effects of UV irradiation duration and H2O2 concentration on the direct oxidation of PFCs and the subsequent GAC adsorption were also explored. The results showed that long-chain perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) consistently exhibited higher removal rates than short-chain perfluorobutyric acid (PFBA) and perfluorobutane sulfonic acid (PFBS) under different process conditions. The removal rates of long chain perfluorinated PFOA and PFOS by GAC adsorption alone were 59.6% and 64.3%, respectively, but the removal rates of short chain perfluorinated PFBA and PFBS were only 11.7% and 13.1%, respectively. When UV or H2O2 were combined with GAC, the removal rates of the four PFCs slightly increased with the increase of UV irradiation time and H2O2 concentration, and the gain effect of UV irradiation was better than that of H2O2.The combined process of UV/H2O2/GAC presented the best removal effect of the four PFCs in water. After 30 minutes of UV/H2O2 treatment, GAC adsorption could remove higher than 90% of PFOA and PFOS, and the removal rates of short chain perfluorinated PFBA and PFBS were over 50%. After 30 min UV/H2O2/GAC treatment, more than 50% PFBA and PFBS were removed, and more than 90% of PFOA and PFOS can be removed. The excellent effect of the UV/H2O2/GAC combined process was due to the hydroxyl radical oxidation reaction generated by the advanced oxidation process of UV/H2O2, which weakened the competitive adsorption of organic matter on PFCs. This study can provide a technical guidance for the treatment of PFCs in water.
-

-
表 1 三维荧光光谱5个分区
Table 1. Five regions of three-dimensional fluorescence spectra
荧光区域 有机物名称 激发波长(Ex)/nm 发射波长(Em)/nm Ⅰ 酪氨酸类 Ex<250 Em<330 Ⅱ 色氨酸类 Ex<250 330<Em<380 Ⅲ 富里酸类 Ex<250 Em>380 Ⅳ 微生物产物类 250<Ex<280 Em<380 Ⅴ 腐殖酸类 Ex>280 Em>380 表 2 目标PFCs的质谱分析条件
Table 2. Target analytes and the MS/MS parameters used in this study
目标物 母离子 子离子 破碎电压/V 碰撞能/V PFBA 213 169.1 57 10 PFBS 299 80 135 32 PFOA 413 368.9 82 4 PFOS 498.85 79.97 80 40 -
[1] LI F, SUN H, HAO Z, et al. Perfluorinated compounds in Haihe River and Dagu drainage canal in Tianjin, China[J]. Chemosphere, 2011, 84(2): 265-271. doi: 10.1016/j.chemosphere.2011.03.060 [2] CHEN S, JIAO X, GAI N, et al. Perfluorinated compounds in soil, surface water, and groundwater from rural areas in eastern China[J]. Environmental Pollution, 2016, 211: 124-131. doi: 10.1016/j.envpol.2015.12.024 [3] YIN H, CHEN R, WANG H, et al. Co-occurrence of phthalate esters and perfluoroalkyl substances affected bacterial community and pathogenic bacteria growth in rural drinking water distribution systems[J]. Science of the Total Environment, 2023, 856: 158943. doi: 10.1016/j.scitotenv.2022.158943 [4] SUN R, WU M, TANG L, et al. Perfluorinated compounds in surface waters of Shanghai, China: Source analysis and risk assessment[J]. Ecotoxicology and Environmental Safety, 2018, 149: 88-95. doi: 10.1016/j.ecoenv.2017.11.012 [5] QI Y, HUO S, HU S, et al. Identification, characterization, and human health risk assessment of perfluorinated compounds in groundwater from a suburb of Tianjin, China[J]. Environmental Earth Sciences, 2016, 75: 1-12. doi: 10.1007/s12665-015-4873-x [6] LI Y, LI J, ZHANG L, et al. Perfluoroalkyl acids in drinking water of China in 2017: distribution characteristics, influencing factors and potential risks[J]. Environment International, 2019, 123: 87-95. doi: 10.1016/j.envint.2018.11.036 [7] SUNDERLAND E M, HU X C, Dassuncao C, et al. A review of the pathways of human exposure to poly-and perfluoroalkyl substances (PFASs) and present understanding of health effects[J]. Journal of Exposure Science & Environmental Epidemiology, 2019, 29(2): 131-147. [8] 孙博, 马军. 水中全氟化合物的活性炭吸附特性研究[J]. 给水排水, 2017, 53(2): 14-18. doi: 10.3969/j.issn.1002-8471.2017.02.003 [9] SON H, KIM T, YOOM H, et al. The adsorption selectivity of short and long per-and polyfluoroalkyl substances (PFASs) from surface water using powder-activated carbon[J]. Water, 2020, 12(11): 3287. doi: 10.3390/w12113287 [10] JAVED H, LYU C, SUN R, et al. Discerning the inefficacy of hydroxyl radicals during perfluorooctanoic acid degradation[J]. Chemosphere, 2020, 247: 125883. doi: 10.1016/j.chemosphere.2020.125883 [11] TANG H, XIANG Q, LEI M, et al. Efficient degradation of perfluorooctanoic acid by UV–Fenton process[J]. Chemical Engineering Journal, 2012, 184: 156-162. doi: 10.1016/j.cej.2012.01.020 [12] CHENG J, LIANG X, YANG S, et al. Photochemical defluorination of aqueous perfluorooctanoic acid(PFOA) by VUV/Fe3+ system[J]. Chemical Engineering Journal, 2014, 239: 242-249. doi: 10.1016/j.cej.2013.11.023 [13] ANUMO T, DAGNINO S, VANDERVORT D R, et al. Transformation of polyfluorinated compounds in natural waters by advanced oxidation processes[J]. Chemosphere, 2016, 144: 1780-1787. doi: 10.1016/j.chemosphere.2015.10.070 [14] YANG S, CHENG J, SUN J, et al. Defluorination of aqueous perfluorooctanesulfonate by activated persulfate oxidation[J]. Plos One, 2013, 8(10): e74877. doi: 10.1371/journal.pone.0074877 [15] 胡晋博, 李梦凯, 严群, 等. 小型化快速柱式反应试验应用于饮用水吸附技术中的研究进展[J]. 给水排水, 2021, 57(3): 17-24. [16] 鲁智礼, 张堯, 黄俊亮, 等. 多相芬顿-活性炭工艺强化饮用水消毒效果[J]. 环境工程学报, 2019, 13(4): 792-799. doi: 10.12030/j.cjee.201812123 [17] MENG P, FANG X, MAIMAITI A, et al. Efficient removal of perfluorinated compounds from water using a regenerable magnetic activated carbon[J]. Chemosphere, 2019, 224: 187-194. doi: 10.1016/j.chemosphere.2019.02.132 [18] GRASSESCHI D, SILVA W C, DE SOUZA PAIVA R, et al. Surface coordination chemistry of graphene: Understanding the coordination of single transition metal atoms[J]. Coordination Chemistry Reviews, 2020, 422: 213469. doi: 10.1016/j.ccr.2020.213469 [19] CAO M H, WANG B B, YU H S, et al. Photochemical decomposition of perfluorooctanoic acid in aqueous periodate with VUV and UV light irradiation[J]. Journal of Hazardous Materials, 2010, 179(1-3): 1143-1146. doi: 10.1016/j.jhazmat.2010.02.030 [20] QU Y, ZHANG C, LI F, et al. Photo-reductive defluorination of perfluorooctanoic acid in water[J]. Water Research, 2010, 44(9): 2939-2947. doi: 10.1016/j.watres.2010.02.019 [21] HORI H, YAMAMOTO A, KOIKE K, et al. Photochemical decomposition of environmentally persistent short-chain perfluorocarboxylic acids in water mediated by iron (II)/(III) redox reactions[J]. Chemosphere, 2007, 68(3): 572-578. doi: 10.1016/j.chemosphere.2006.12.038 [22] JING C, ZHANG P, JIAN L I U. Photodegradation of perfluorooctanoic acid by 185 nm vacuum ultraviolet light[J]. Journal of Environmental Sciences, 2007, 19(4): 387-390. doi: 10.1016/S1001-0742(07)60064-3 [23] GIRI R R, OZAKI H, OKADA T, et al. Water matrix effect on UV photodegradation of perfluorooctanoic acid[J]. Water Science and Technology, 2011, 64(10): 1980-1986. doi: 10.2166/wst.2011.825 [24] MATILAINEN A, SILAN M. Removal of natural organic matter from drinking water by advanced oxidation processes[J]. Chemosphere, 2010, 80(4): 351-365. doi: 10.1016/j.chemosphere.2010.04.067 [25] WANG G S, LIAO C H, CHEN H W, et al. Characteristics of natural organic matter degradation in water by UV/H2O2 treatment[J]. Environmental Technology, 2006, 27(3): 277-287. doi: 10.1080/09593332708618638 [26] LIU X, WEI W, XU J, et al. Photochemical decomposition of perfluorochemicals in contaminated water[J]. Water Research, 2020, 186: 116311. doi: 10.1016/j.watres.2020.116311 [27] CHEN Z, TENG Y, MI N, et al. Highly efficient hydrated electron utilization and reductive destruction of perfluoroalkyl substances induced by intermolecular interaction[J]. Environmental Science & Technology, 2021, 55(6): 3996-4006. [28] PARK H, VECITIS C D, CHENG J, et al. Reductive defluorination of aqueous perfluorinated alkyl surfactants: Effects of ionic headgroup and chain length[J]. The Journal of Physical Chemistry A, 2009, 113(4): 690-696. doi: 10.1021/jp807116q [29] PARKINSON A, RODDICK F A, HOBDAY M D. UV photooxidation of NOM: issues related to drinking water treatment[J]. Journal of Water Supply:Research and Technology—AQUA, 2003, 52(8): 577-586. doi: 10.2166/aqua.2003.0051 [30] YU J, LV L, LAN P, et al. Effect of effluent organic matter on the adsorption of perfluorinated compounds onto activated carbon[J]. Journal of Hazardous Materials, 2012, 225: 99-106. [31] MATSUI Y, KNAPPE D R, IWAKI K, et al. Pesticide adsorption by granular activated carbon adsorbers. 2. Effects of pesticide and natural organic matter characteristics on pesticide breakthrough curves[J]. Environmental Science & Technology, 2002, 36(15): 3432-3438. [32] MATAFONVA G, BATOEV V. Recent advances in application of UV light-emitting diodes for degrading organic pollutants in water through advanced oxidation processes: A review[J]. Water Research, 2018, 132: 177-189. doi: 10.1016/j.watres.2017.12.079 [33] APPLEMAN T D, DICKENSON E R, BELLONA C, et al. Nanofiltration and granular activated carbon treatment of perfluoroalkyl acids[J]. Journal of Hazardous Materials, 2013, 260: 740-746. doi: 10.1016/j.jhazmat.2013.06.033 [34] MCCLEAF P, ENFLUND S, östlund A, et al. Removal efficiency of multiple poly-and perfluoroalkyl substances (PFASs) in drinking water using granular activated carbon (GAC) and anion exchange (AE) column tests[J]. Water Research, 2017, 120: 77-87. doi: 10.1016/j.watres.2017.04.057 [35] SGROI M, ANUMOLT, ROCCARO P, et al. Modeling emerging contaminants breakthrough in packed bed adsorption columns by UV absorbance and fluorescing components of dissolved organic matter[J]. Water Research, 2018, 145: 667-677. doi: 10.1016/j.watres.2018.09.018 -



 下载:
下载: